What Is a User Requirement Specification (URS)? How to Write and Manage One
The Essential Guide to Requirements Management and Traceability
Chapters
- 1. Requirements Management
- Overview
- 1 What is Requirements Management? A Complete Guide
- 2 Why do you need Requirements Management?
- 3 Four Stages of Requirements Management Processes
- 4 Adopting an Agile Approach to Requirements Management
- 5 Status Request Changes
- 6 Conquering the 5 Biggest Challenges of Requirements Management
- 7 Three Reasons You Need a Requirements Management Solution
- 8 Guide to Poor Requirements: Identify Causes, Repercussions, and How to Fix Them
- 2. Writing Requirements
- Overview
- 1 Functional requirements examples and templates
- 2 What Is a Product Requirements Document? A Complete PRD Guide
- 3 What Is a User Requirement Specification (URS)? How to Write and Manage One
- 4 Identifying and Measuring Requirements Quality
- 5 How to Write a System Requirements Specification (SRS) Document
- 6 The Fundamentals of Business Requirements: Examples of Business Requirements and the Importance of Excellence
- 7 What Is a Compliance Risk Assessment? Steps, Framework, and Examples
- 8 Adopting the EARS Notation to Improve Requirements Engineering
- 9 Jama Connect Advisor™
- 10 Frequently Asked Questions about the EARS Notation and Jama Connect Advisor™
- 11 How to Write an Effective Product Requirements Document (PRD)
- 12 Functional vs. Non-Functional Requirements
- 13 What Are Nonfunctional Requirements and How Do They Impact Product Development?
- 14 What Is a Software Design Specification? Key Components + Template
- 15 Characteristics of Effective Software Requirements and Software Requirements Specifications (SRS)
- 16 8 Do’s and Don’ts for Writing Requirements
- 17 Project Requirements: Types, Process, and Best Practices
- 3. Requirements Gathering and Management Processes
- Overview
- 1 Requirements Engineering
- 2 Requirements Analysis
- 3 A Guide to Requirements Elicitation for Product Teams
- 4 Requirements Gathering Techniques for Agile Product Teams
- 5 Requirements Gathering in Software Engineering: Process, Techniques, and Best Practices
- 6 Defining and Implementing a Requirements Baseline
- 7 Managing Project Scope — Why It Matters and Best Practices
- 8 How Long Do Requirements Take?
- 9 How to Reuse Requirements Across Multiple Products
- 4. Requirements Traceability
- Overview
- 1 What Is Traceability in Product Development? A Guide for Regulated Teams
- 2 Tracing Your Way to Success: The Crucial Role of Traceability in Modern Product and Systems Development
- 3 Bidirectional Traceability: What It Is and How to Implement It
- 4 What is Engineering Change Management (ECM)? A Complete Guide
- 5 Change Impact Analysis (CIA): A Short Guide for Effective Implementation
- 6 What is Meant by Version Control?
- 7 Key Traceability Challenges and Tips for Ensuring Accountability and Efficiency
- 8 The Role of a Data Thread in Product and Software Development
- 9 Unraveling the Digital Thread: Enhancing Connectivity and Efficiency
- 10 What is a Traceability Matrix? A Guide to Requirements Traceability
- 11 How to Create and Use a Requirements Traceability Matrix (RTM)
- 12 Requirements Traceability Matrix Pros and Cons: A Practical Guide
- 13 Live Traceability vs. After-the-Fact Traceability
- 14 Overcoming Barriers to Live Requirements Traceability™
- 15 Requirements Traceability, What Are You Missing?
- 16 Requirements Traceability: Links in the Chain
- 17 What Are the Benefits of End-to-End Traceability During Product Development?
- 18 FAQs About Requirements Traceability
- 19 Product Traceability for Regulated Industries: A Complete Guide to Audit-Ready Compliance
- 5. Requirements Management Tools and Software
- Overview
- 1 Selecting the Right Requirements Management Tools and Software
- 2 Why Investing in Requirements Management Software Makes Business Sense During an Economic Downturn
- 3 Why Word and Excel Alone is Not Enough for Product, Software, and Systems Development
- 4 Can You Track Requirements in Excel?
- 5 What Is Application Lifecycle Management (ALM)?
- 6 Is There Life After DOORS®?
- 7 Can You Track Requirements in Jira?
- 8 Checklist: Selecting a Requirements Management Tool
- 6. Requirements Validation and Verification
- 7. Meeting Regulatory Compliance and Industry Standards
- Overview
- 1 Understanding ISO Standards
- 2 Understanding ISO/IEC 27001: A Guide to Information Security Management
- 3 What is DevSecOps? A Guide to Building Secure Software
- 4 Compliance Management
- 5 What Is Functional Safety (FuSa)? Standards, Lifecycle, and Where Programs Fail
- 6 What is FMEA? Failure Mode and Effects Analysis Guide
- 7 TÜV SÜD: Ensuring Safety, Quality, and Sustainability Worldwide
- 8 What is IEC 62443? A Guide to Industrial Cybersecurity
- 8. Systems Engineering
- Overview
- 1 What is Systems Engineering?
- 2 How Do Engineers Collaborate? A Guide to Streamlined Teamwork and Innovation
- 3 The Systems Engineering Body of Knowledge (SEBoK)
- 4 What Is MBSE? Model-Based Systems Engineering Explained
- 5 Digital Engineering Between Government and Contractors
- 6 Digital Engineering Tools: The Key to Driving Innovation and Efficiency in Complex Systems
- 9. Automotive Development
- Overview
- 1 Understanding IATF 16949: A Quick Guide to Automotive Quality Management
- 2 What Is ISO 21434? Automotive Cybersecurity Engineering Explained
- 3 What Is ISO 26262? A Guide to Functional Safety in Automotive
- 4 What Is ASIL? A Guide to Automotive Safety Integrity Levels in ISO 26262
- 5 What Is SOTIF? A Guide to ISO 21448 for ADAS Safety
- 10. Medical Device & Life Sciences Development
- Overview
- 1 The Importance of Benefit-Risk Analysis in Medical Device Development
- 2 Software as a Medical Device: Revolutionizing Healthcare
- 3 What’s a Design History File, and How Are DHFs Used by Product Teams?
- 4 Navigating the Risks of Software of Unknown Pedigree (SOUP) in the Medical Device & Life Sciences Industry
- 5 What Is ISO 13485? A Guide to Medical Device Quality Management Systems
- 6 What You Need to Know: ANSI/AAMI SW96:2023 — Medical Device Security
- 7 ISO 13485 vs ISO 9001: Understanding the Differences and Synergies
- 8 What Is IEC 62304? A Guide to Medical Device Software
- 9 What Is a Device Master Record (DMR)? Definition and FDA Requirements
- 10 Failure Modes, Effects, and Diagnostic Analysis (FMEDA) for Medical Devices: What You Need to Know
- 11 Embracing the Future of Healthcare: Exploring the Internet of Medical Things (IoMT)
- 12 What Is General Safety and Performance Requirements (GSPR)? What You Need To Know
- 13 What Is IEC 62366? A Guide to Medical Device Usability Engineering
- 11. Aerospace & Defense Development
- Overview
- 1 What Is ARP4754A? A Complete Guide to Civil Aircraft and Systems Development Assurance
- 2 Understanding ARP4761A: Guidelines for System Safety Assessment in Aerospace
- 3 What Is DO-254? A Complete Guide to Airborne Hardware Design Assurance
- 4 What Is DO-178C? A Complete Guide to Airborne Software Certification
- 12. Architecture, Engineering, and Construction (AEC industry) Development
- 13. Industrial Manufacturing & Machinery, Automation & Robotics, Consumer Electronics, and Energy
- 14. Semiconductor Development
- 15. AI in Product Development
- Overview
- 1 What Is AI in Product Development? A Complete 2026 Guide
- 2 AI Test Case Generation: A Complete Guide for Regulated QA Teams
- 3 Using AI to Write Software Requirements: What Works and What Doesn’t
- 4 What Is the Model Context Protocol (MCP) for Requirements Management?
- 5 AI for Systems Engineering: Benefits, Risks, and How to Start
- 6 Artificial Intelligence in Requirements Management
- 16. Risk Management
- 17. Product Development Terms and Definitions
Chapter 2: What Is a User Requirement Specification (URS)? How to Write and Manage One
Chapters
- 1. Requirements Management
- Overview
- 1 What is Requirements Management? A Complete Guide
- 2 Why do you need Requirements Management?
- 3 Four Stages of Requirements Management Processes
- 4 Adopting an Agile Approach to Requirements Management
- 5 Status Request Changes
- 6 Conquering the 5 Biggest Challenges of Requirements Management
- 7 Three Reasons You Need a Requirements Management Solution
- 8 Guide to Poor Requirements: Identify Causes, Repercussions, and How to Fix Them
- 2. Writing Requirements
- Overview
- 1 Functional requirements examples and templates
- 2 What Is a Product Requirements Document? A Complete PRD Guide
- 3 What Is a User Requirement Specification (URS)? How to Write and Manage One
- 4 Identifying and Measuring Requirements Quality
- 5 How to Write a System Requirements Specification (SRS) Document
- 6 The Fundamentals of Business Requirements: Examples of Business Requirements and the Importance of Excellence
- 7 What Is a Compliance Risk Assessment? Steps, Framework, and Examples
- 8 Adopting the EARS Notation to Improve Requirements Engineering
- 9 Jama Connect Advisor™
- 10 Frequently Asked Questions about the EARS Notation and Jama Connect Advisor™
- 11 How to Write an Effective Product Requirements Document (PRD)
- 12 Functional vs. Non-Functional Requirements
- 13 What Are Nonfunctional Requirements and How Do They Impact Product Development?
- 14 What Is a Software Design Specification? Key Components + Template
- 15 Characteristics of Effective Software Requirements and Software Requirements Specifications (SRS)
- 16 8 Do’s and Don’ts for Writing Requirements
- 17 Project Requirements: Types, Process, and Best Practices
- 3. Requirements Gathering and Management Processes
- Overview
- 1 Requirements Engineering
- 2 Requirements Analysis
- 3 A Guide to Requirements Elicitation for Product Teams
- 4 Requirements Gathering Techniques for Agile Product Teams
- 5 Requirements Gathering in Software Engineering: Process, Techniques, and Best Practices
- 6 Defining and Implementing a Requirements Baseline
- 7 Managing Project Scope — Why It Matters and Best Practices
- 8 How Long Do Requirements Take?
- 9 How to Reuse Requirements Across Multiple Products
- 4. Requirements Traceability
- Overview
- 1 What Is Traceability in Product Development? A Guide for Regulated Teams
- 2 Tracing Your Way to Success: The Crucial Role of Traceability in Modern Product and Systems Development
- 3 Bidirectional Traceability: What It Is and How to Implement It
- 4 What is Engineering Change Management (ECM)? A Complete Guide
- 5 Change Impact Analysis (CIA): A Short Guide for Effective Implementation
- 6 What is Meant by Version Control?
- 7 Key Traceability Challenges and Tips for Ensuring Accountability and Efficiency
- 8 The Role of a Data Thread in Product and Software Development
- 9 Unraveling the Digital Thread: Enhancing Connectivity and Efficiency
- 10 What is a Traceability Matrix? A Guide to Requirements Traceability
- 11 How to Create and Use a Requirements Traceability Matrix (RTM)
- 12 Requirements Traceability Matrix Pros and Cons: A Practical Guide
- 13 Live Traceability vs. After-the-Fact Traceability
- 14 Overcoming Barriers to Live Requirements Traceability™
- 15 Requirements Traceability, What Are You Missing?
- 16 Requirements Traceability: Links in the Chain
- 17 What Are the Benefits of End-to-End Traceability During Product Development?
- 18 FAQs About Requirements Traceability
- 19 Product Traceability for Regulated Industries: A Complete Guide to Audit-Ready Compliance
- 5. Requirements Management Tools and Software
- Overview
- 1 Selecting the Right Requirements Management Tools and Software
- 2 Why Investing in Requirements Management Software Makes Business Sense During an Economic Downturn
- 3 Why Word and Excel Alone is Not Enough for Product, Software, and Systems Development
- 4 Can You Track Requirements in Excel?
- 5 What Is Application Lifecycle Management (ALM)?
- 6 Is There Life After DOORS®?
- 7 Can You Track Requirements in Jira?
- 8 Checklist: Selecting a Requirements Management Tool
- 6. Requirements Validation and Verification
- 7. Meeting Regulatory Compliance and Industry Standards
- Overview
- 1 Understanding ISO Standards
- 2 Understanding ISO/IEC 27001: A Guide to Information Security Management
- 3 What is DevSecOps? A Guide to Building Secure Software
- 4 Compliance Management
- 5 What Is Functional Safety (FuSa)? Standards, Lifecycle, and Where Programs Fail
- 6 What is FMEA? Failure Mode and Effects Analysis Guide
- 7 TÜV SÜD: Ensuring Safety, Quality, and Sustainability Worldwide
- 8 What is IEC 62443? A Guide to Industrial Cybersecurity
- 8. Systems Engineering
- Overview
- 1 What is Systems Engineering?
- 2 How Do Engineers Collaborate? A Guide to Streamlined Teamwork and Innovation
- 3 The Systems Engineering Body of Knowledge (SEBoK)
- 4 What Is MBSE? Model-Based Systems Engineering Explained
- 5 Digital Engineering Between Government and Contractors
- 6 Digital Engineering Tools: The Key to Driving Innovation and Efficiency in Complex Systems
- 9. Automotive Development
- Overview
- 1 Understanding IATF 16949: A Quick Guide to Automotive Quality Management
- 2 What Is ISO 21434? Automotive Cybersecurity Engineering Explained
- 3 What Is ISO 26262? A Guide to Functional Safety in Automotive
- 4 What Is ASIL? A Guide to Automotive Safety Integrity Levels in ISO 26262
- 5 What Is SOTIF? A Guide to ISO 21448 for ADAS Safety
- 10. Medical Device & Life Sciences Development
- Overview
- 1 The Importance of Benefit-Risk Analysis in Medical Device Development
- 2 Software as a Medical Device: Revolutionizing Healthcare
- 3 What’s a Design History File, and How Are DHFs Used by Product Teams?
- 4 Navigating the Risks of Software of Unknown Pedigree (SOUP) in the Medical Device & Life Sciences Industry
- 5 What Is ISO 13485? A Guide to Medical Device Quality Management Systems
- 6 What You Need to Know: ANSI/AAMI SW96:2023 — Medical Device Security
- 7 ISO 13485 vs ISO 9001: Understanding the Differences and Synergies
- 8 What Is IEC 62304? A Guide to Medical Device Software
- 9 What Is a Device Master Record (DMR)? Definition and FDA Requirements
- 10 Failure Modes, Effects, and Diagnostic Analysis (FMEDA) for Medical Devices: What You Need to Know
- 11 Embracing the Future of Healthcare: Exploring the Internet of Medical Things (IoMT)
- 12 What Is General Safety and Performance Requirements (GSPR)? What You Need To Know
- 13 What Is IEC 62366? A Guide to Medical Device Usability Engineering
- 11. Aerospace & Defense Development
- Overview
- 1 What Is ARP4754A? A Complete Guide to Civil Aircraft and Systems Development Assurance
- 2 Understanding ARP4761A: Guidelines for System Safety Assessment in Aerospace
- 3 What Is DO-254? A Complete Guide to Airborne Hardware Design Assurance
- 4 What Is DO-178C? A Complete Guide to Airborne Software Certification
- 12. Architecture, Engineering, and Construction (AEC industry) Development
- 13. Industrial Manufacturing & Machinery, Automation & Robotics, Consumer Electronics, and Energy
- 14. Semiconductor Development
- 15. AI in Product Development
- Overview
- 1 What Is AI in Product Development? A Complete 2026 Guide
- 2 AI Test Case Generation: A Complete Guide for Regulated QA Teams
- 3 Using AI to Write Software Requirements: What Works and What Doesn’t
- 4 What Is the Model Context Protocol (MCP) for Requirements Management?
- 5 AI for Systems Engineering: Benefits, Risks, and How to Start
- 6 Artificial Intelligence in Requirements Management
- 16. Risk Management
- 17. Product Development Terms and Definitions
What Is a User Requirement Specification (URS)? How to Write and Manage One
A user requirement specification (URS) gives every team the same interpretation of what a system needs to do, and gives every auditor a single thread to trace from user need through design, test, and validation. When it’s written before anyone picks a vendor or writes a line of code, it protects the project from scope drift, misaligned designs, and preventable audit findings.
This guide covers what a URS is, how it differs from other specification documents, how to write requirements that hold up to regulatory scrutiny, and how to manage the URS after it’s baselined.
What Is a User Requirement Specification?
A user requirement specification (URS) defines what a system must do, based on business needs and written without locking in any specific technology. It captures user needs and intended use before the engineering team decides how to build anything, and a properly written URS stays valid regardless of which vendor or platform you choose.
The URS carries formal standing in regulated work. Under Good Manufacturing Practice (GMP) Annex 11, user requirements must remain traceable throughout the lifecycle, meaning you can follow each requirement from its origin through design, testing, and final validation. GAMP 5 guidance uses the URS as the reference point for Performance Qualification (PQ), where teams prove the finished system actually meets user needs.
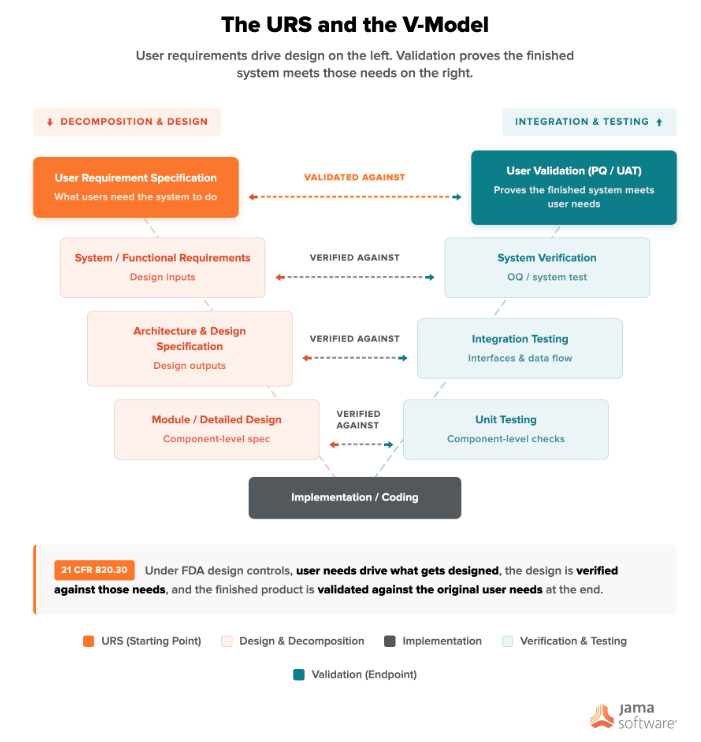
Where the URS Fits in the Development Lifecycle
The URS is the starting point of the V-model. It sits at the top left, and the validation activities that prove the system works sit directly across on the right. Under FDA design controls (21 CFR 820.30), user needs drive what gets designed, the design is verified against those needs, and the finished product is validated against the original user needs at the end.
The URS must be completed before you select a vendor, define contracts, or begin qualification testing. A URS written after procurement misses the point because it no longer influences the design.
How the URS Differs From an SRS and FRD
The URS, software requirements specification (SRS), and functional requirements document (FRD) sit at different levels. The URS describes the outcome in the user’s language. The SRS defines how the software will work, down to parameter names, error codes, and timing. The FRD describes specific system behaviors tied to a chosen technology.
For example, a URS for an insulin pump might say “prevent bolus doses exceeding the clinician-set maximum.” The SRS would then specify that the software checks each dose against a stored limit, rejects entries over it, and displays an error code within 500 milliseconds. Keeping these layers separate is a compliance requirement under both FDA guidance and the IEEE 29148 standard.
Core Components of a URS
A regulated URS organizes requirements into categories, and each category has its own rules for how you prove the requirement was met. Every requirement needs measurable conditions so it can actually be tested.
Functional Requirements
Functional requirements describe what the system must do: its behaviors, inputs, outputs, and capabilities. Each one describes an action, not the mechanism behind it. In a regulated URS, every functional requirement gets a unique ID and description. It may also get a criticality level that indicates how important it is.
Regulated URS templates commonly use hierarchical codes like UR.F001.01, where the prefix tells you the category and the numbers narrow down to the specific sub-requirement. Any requirement tied to Good Practice (GxP) standards, the umbrella term for quality regulations in pharma and life sciences, gets marked as “MUST” per ISPE GAMP 5 templates.
Non-Functional Requirements
How fast, how available, and how usable a system needs to be is just as auditable as what it does. Non-functional requirements cover quality attributes and operational constraints like response time, uptime, and usability. A user need like “Portable” isn’t testable on its own. You need to turn it into something measurable, like “weighs no more than 5 lbs plus or minus 1 lb.” When non-functional requirements conflict with each other, FDA design controls guidance requires that you resolve and document the tradeoff.
Safety and Regulatory Requirements
Safety requirements describe the conditions the system must meet to prevent harm. They come from hazard analysis, where the team identifies what could go wrong and how severe it would be. The findings from that analysis feed into design inputs, and safety controls that come out of it may be written directly into the URS. Regulatory requirements define the compliance obligations that laws, standards bodies, or regulatory authorities impose on your system. A bioprocess equipment URS, for example, might require that lubricants in contact with process fluid comply with 21 CFR Part 178 and be certified to United States Department of Agriculture (USDA) H1 or equivalent.
Acceptance Criteria
Without a clear pass/fail line, you can’t verify a requirement. Acceptance criteria are the measurable conditions a requirement must satisfy to pass. They have to be objective and set before testing begins.
The FDA is specific about the word “predetermined.” You define what “pass” looks like before you run the test, not after you see the results. A well-formed acceptance criterion covers four elements:
- Parameter: What the test evaluates.
- Measurement method: How the team measures the parameter.
- Pass/fail threshold: The limit that determines success or failure.
- Test conditions: The conditions under which the result is valid.
Vague acceptance criteria create gaps that show up as audit findings later.
How to Write a User Requirement Specification
Writing a URS that survives regulatory scrutiny follows a sequence. Gather needs broadly, write at the right level, make every requirement testable, then lock the document through formal review.
1. Gather Needs From All Contributing Teams
Requirements elicitation, the process of gathering what stakeholders actually need, requires input from every group that touches the system. A URS written by a single engineer and rubber-stamped by management is a documented failure mode. Common contributor groups include:
- Production and operations staff: Define how the system needs to work in daily use.
- Quality assurance leads: Review for compliance needs and validation expectations.
- Engineering and maintenance teams: Contribute technical constraints and serviceability needs.
- IT and systems engineering groups: Provide input on how the system fits into the broader environment.
- Regulatory affairs: Identify compliance obligations that must appear in the URS.
- Suppliers: Provide constraints or interface needs that affect the final requirements.
The more complex or risky the system, the more deeply each group should be involved.
2. Write at the User Level, Not the Solution Level
If a developer reads a requirement and has zero design freedom because the requirement already specifies the solution, it’s written at the wrong level. A good format to follow: [who] shall [do what] [to what] [under what conditions]. The word “shall” signals a binding requirement. Each requirement should contain one statement per sentence, use active voice, and avoid stacking multiple conditions into one clause.
“The aircraft shall have three engines” prescribes a design. “The aircraft shall meet the operation requirements with a single engine out” defines a capability and leaves the design team free to determine how to achieve it.
3. Make Each Requirement Testable and Unambiguous
Every requirement should be necessary, appropriate, unambiguous, complete, and singular. Those quality checks come from INCOSE’s Guide to Writing Requirements, a widely used reference for writing testable requirements. Vague terms like “fast,” “easy,” or “user-friendly” fail because they can’t be measured or tested consistently.
The Easy Approach to Requirements Syntax (EARS) gives you sentence templates that turn vague language into testable statements. An ambiguous requirement like “the system should handle errors” becomes testable when rewritten as “If the radar fails, then the cruise control system shall enter standby mode.”
4. Obtain Formal Review and Sign-Off
Cross-functional review is the last checkpoint before the URS is locked in as the baseline. Reviewers check three things: every requirement traces back to a real stated need, acceptance criteria are defined for each requirement, and the URS as a whole reflects actual operational needs. Once the document is signed off, any changes require formal approval, a documented impact assessment, and an updated version number.
Managing the URS in Regulated Industries
Once approved and baselined, the URS becomes the reference point for configuration management, traceability, and validation. Every change to a baselined requirement ripples downstream. Design documents, test protocols, and risk assessments that link to that requirement all need to be reviewed.
Maintaining Bidirectional Traceability
Bidirectional traceability is a core expectation in regulated work. Forward traceability means you can follow a requirement from the URS through design and into testing. Most teams build this naturally. Backward traceability means you can start at any test and trace it back through design to the original requirement. Reviewers use backward tracing to confirm that no test exists without a corresponding requirement and no requirement exists without evidence that it was verified.
Requirements that don’t apply still need to be called out, justified, and recorded in the traceability matrix. If a requirement is silently absent from the matrix, a reviewer can’t tell whether it was intentionally excluded or accidentally missed.
Controlling Post-Baseline Changes
Once the baseline is set, you can’t just edit the document. Every change goes through formal change control. Each approved URS change triggers updates across four areas:
- Traceability matrix: The matrix must reflect the revised requirement and its links.
- Risk assessment: Linked risk assessments need re-evaluation.
- Test protocols: Affected protocols must be updated and re-executed where needed.
- SOPs and training: Standard operating procedures and training materials tied to changed requirements must be revised.
Skip any one of these downstream updates, and you’ve created a gap. Auditors treat it the same way they treat a missing requirement. The traceability chain is broken either way.
How Jama Connect Supports URS Management
Jama Connect® is a requirements management platform that keeps URS requirements, design elements, test cases, and risk items connected in a single system. When you manage a URS manually across spreadsheets and shared drives, things break down as projects grow. A requirement changes in one file, but the linked test protocol in another file doesn’t get updated, and nobody notices until an auditor pulls the thread.
With Jama Connect, suspect-link flags automatically tell downstream owners exactly what needs review the moment something changes. Teams can see the full trace from any URS requirement through to its design output, verification activity, and risk assessment. That visibility means you catch gaps in real time instead of scrambling to reconstruct traceability before an audit or submission deadline.
Turning URS Compliance Into a Continuous Workflow
The hardest part of URS management isn’t writing the initial document. It’s keeping everything downstream, design documents, test protocols, risk assessments, aligned as requirements change due to design updates, new regulations, or risk findings that surface after the baseline is set. Teams that build traceability into their daily workflow, connecting every requirement to design evidence and reviewing downstream artifacts whenever something changes, spend less time preparing for audits and more time building. Start a free 30-day trial of Jama Connect to see how it fits your process.
Frequently Asked Questions About User Requirement Specification (URS)
What is the difference between a URS and an SRS?
A URS describes what users need the system to do, in their language, without specifying how the software should work. An SRS translates those user-level needs into software-specific detail: parameter names, error codes, timing constraints, and testable thresholds. Both are required in regulated development, but the URS must exist first because it defines the acceptance criteria that validation ultimately confirms.
Who is responsible for writing a URS?
No single role owns the URS. The system or equipment owner typically leads the writing. End users define functional and operational requirements. Quality assurance reviews for regulatory compliance and gives final approval. Engineering provides input on what’s technically feasible. The more complex or risky the system, the more deeply each group should be involved.
How detailed should a URS be?
Detailed enough that every requirement is testable and unambiguous, but not so detailed that it prescribes the solution. GAMP 5 helps you calibrate the right level of detail based on risk, complexity, and how new the system is. For custom software (Category 4 and 5 systems in GAMP terms), requirements should be developed without assuming a specific solution. Requirements related to patient safety, product quality, and data integrity should always be specified, no matter what category the system falls into.
Does a URS need to be updated after a project begins?
Yes, but only through formal change control. The URS should stay current throughout development because design, testing, and validation all trace back to it. Once it’s baselined, adding, deleting, or modifying any requirement requires formal approval, a documented assessment of what the change affects downstream, and an updated version number. Jama Connect automates the impact assessment step by flagging every linked artifact when a baselined requirement changes.
Book a Demo
See Jama Connect in Action!
Our Jama Connect experts are ready to guide you through a personalized demo, answer your questions, and show you how Jama Connect can help you identify risks, improve cross-team collaboration, and drive faster time to market.