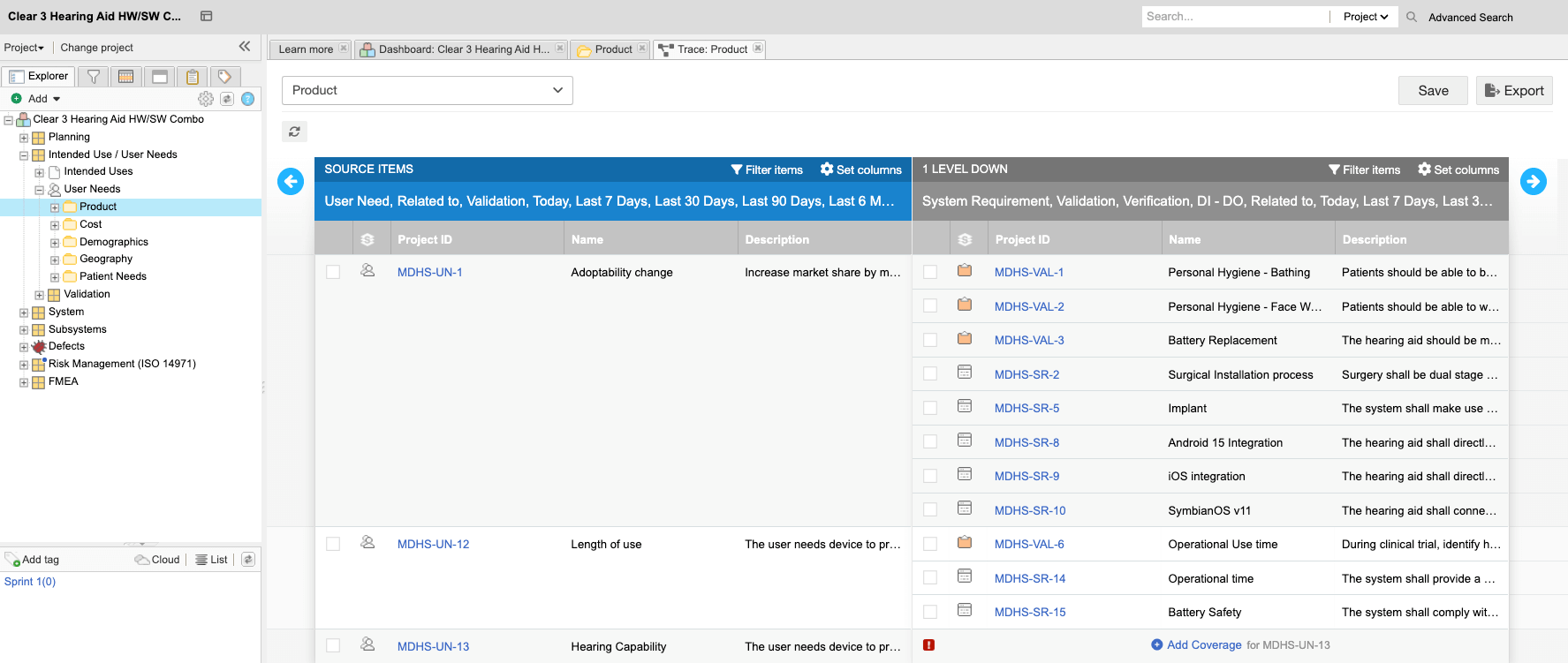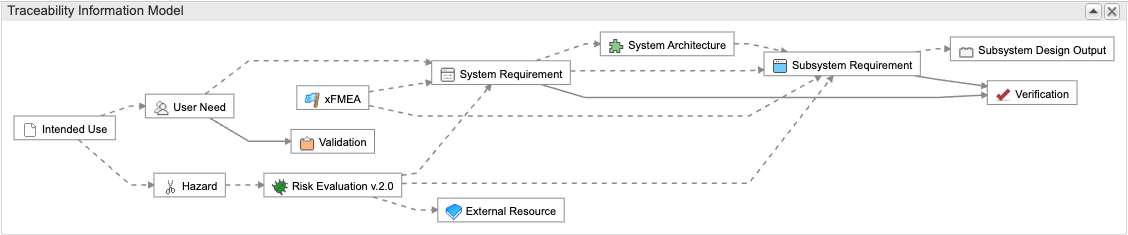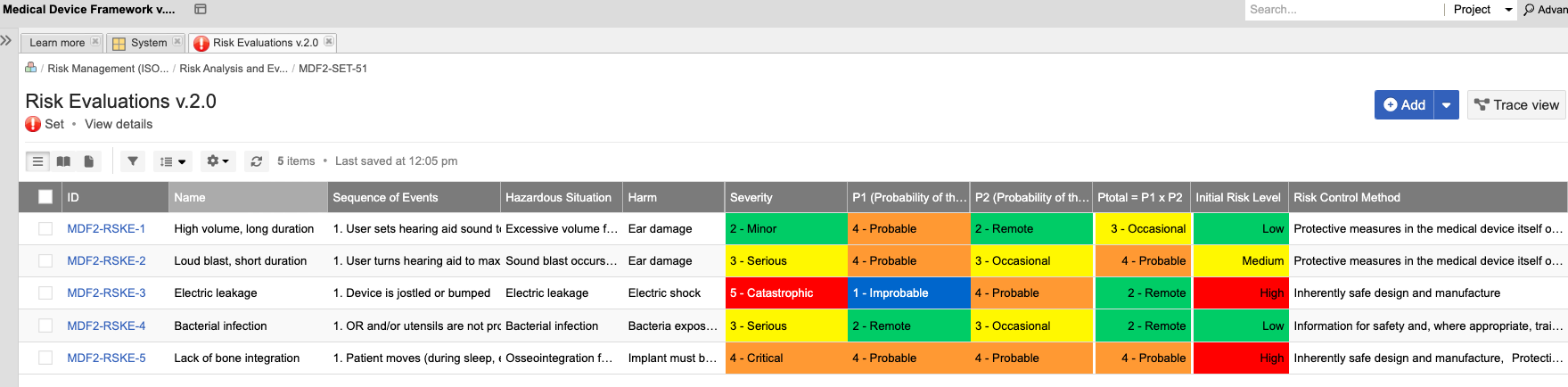
Engineering Management Platform for Faster, Safer
MedTech & Life Sciences Development
Trusted by the World’s Leading Companies
How High-Performing Engineering Teams Use Jama Connect to Accelerate Development
60% Efficiency Improvement
“Other tools have traceability, but only Jama Connect has a visual, configurable model that shows exactly what traceability looks like and accommodates our product’s complex configuration.”
Sri Harsha Maramraju
Senior Manager, Systems Design Engineering

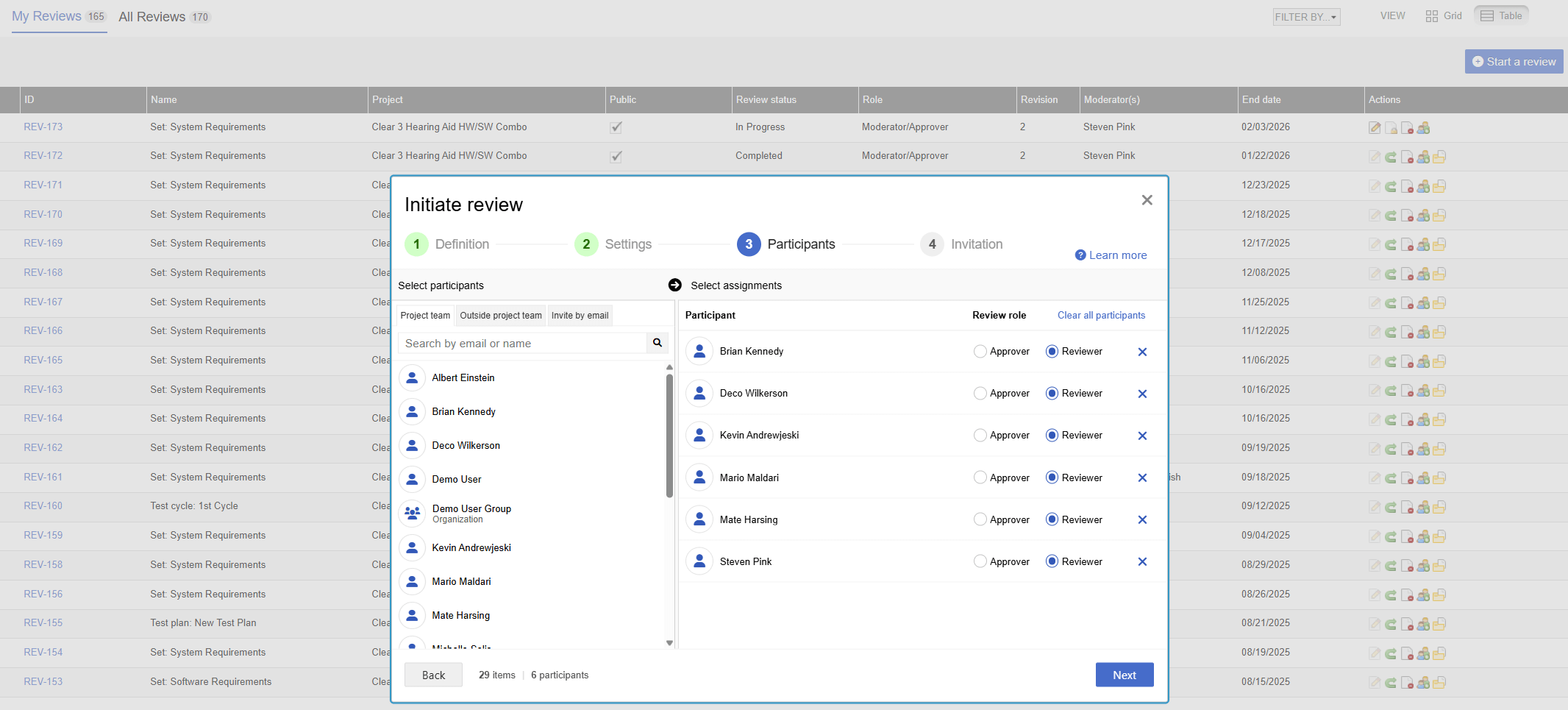
Trace Matrices Generation in 1 Day
“Jama Connect’s automation allows us to efficiently manage each release. The tool allows us to reduce manual processes involved in generating traceability and focus our efforts on reviews of the trace matrix. Efficiency and speed are critical in managing our software cadence and to be competitive in the marketplace.”
Sandhya Mitnala
Head of Quality and Regulatory

80 Hours per Project Saved
“With the Review Center, we review things more incrementally, and we can catch changes, or things to fix, very early in the process. So, the cost at the end is less.”
Carmen Pazos
Diagnostic Divisions R&D, Instruments Sr Manager

We Offer the Best Support for Every Stage and Size
With Jama Connect, companies report:
28%
Average decrease in time for traceability
23%
Average increase in use of requirements
21%
Average reduction in audit preparations
17%
Average decrease in review cycle times
Stop Drowning in Documentation.
Start Accelerating Compliance and Innovation.
One Platform. All Products. Total Confidence.
Enterprise-Grade Scalability, Security and Reliability
Frequently Asked Questions
See Jama Connect in Action!
Our Jama Connect experts are ready to guide you through a personalized demo for requirements management for medical device development, answer your questions, and show you how
Jama Connect can help you identify risks, improve cross-team collaboration, and drive faster time to market through AI-native engineering management.