What is Requirements Management? A Complete Guide
The Essential Guide to Requirements Management and Traceability
Chapters
- 1. Requirements Management
- Overview
- 1 What is Requirements Management? A Complete Guide
- 2 Why do you need Requirements Management?
- 3 Four Stages of Requirements Management Processes
- 4 Adopting an Agile Approach to Requirements Management
- 5 Status Request Changes
- 6 Conquering the 5 Biggest Challenges of Requirements Management
- 7 Three Reasons You Need a Requirements Management Solution
- 8 Guide to Poor Requirements: Identify Causes, Repercussions, and How to Fix Them
- 2. Writing Requirements
- Overview
- 1 Functional requirements examples and templates
- 2 What Is a Product Requirements Document? A Complete PRD Guide
- 3 Identifying and Measuring Requirements Quality
- 4 What Is a User Requirement Specification (URS)? How to Write and Manage One
- 5 How to Write a System Requirements Specification (SRS) Document
- 6 The Fundamentals of Business Requirements: Examples of Business Requirements and the Importance of Excellence
- 7 Adopting the EARS Notation to Improve Requirements Engineering
- 8 What Is a Compliance Risk Assessment? Steps, Framework, and Examples
- 9 Jama Connect Advisor™
- 10 Frequently Asked Questions about the EARS Notation and Jama Connect Advisor™
- 11 How to Write an Effective Product Requirements Document (PRD)
- 12 Functional vs. Non-Functional Requirements
- 13 What Are Nonfunctional Requirements and How Do They Impact Product Development?
- 14 Characteristics of Effective Software Requirements and Software Requirements Specifications (SRS)
- 15 What Is a Software Design Specification? Key Components + Template
- 16 8 Do’s and Don’ts for Writing Requirements
- 3. Requirements Gathering and Management Processes
- Overview
- 1 Requirements Engineering
- 2 Requirements Analysis
- 3 A Guide to Requirements Elicitation for Product Teams
- 4 Requirements Gathering Techniques for Agile Product Teams
- 5 Requirements Gathering in Software Engineering: Process, Techniques, and Best Practices
- 6 Defining and Implementing a Requirements Baseline
- 7 Managing Project Scope — Why It Matters and Best Practices
- 8 How Long Do Requirements Take?
- 9 How to Reuse Requirements Across Multiple Products
- 4. Requirements Traceability
- Overview
- 1 How is Traceability Achieved? A Practical Guide for Engineers
- 2 What is Requirements Traceability? Importance Explained
- 3 Tracing Your Way to Success: The Crucial Role of Traceability in Modern Product and Systems Development
- 4 Bidirectional Traceability: What It Is and How to Implement It
- 5 What is Engineering Change Management (ECM)? A Complete Guide
- 6 Change Impact Analysis (CIA): A Short Guide for Effective Implementation
- 7 What is Meant by Version Control?
- 8 What is Requirements Traceability and Why Does It Matter for Product Teams?
- 9 Key Traceability Challenges and Tips for Ensuring Accountability and Efficiency
- 10 The Role of a Data Thread in Product and Software Development
- 11 Unraveling the Digital Thread: Enhancing Connectivity and Efficiency
- 12 What is a Traceability Matrix? A Guide to Requirements Traceability
- 13 How to Create and Use a Requirements Traceability Matrix (RTM)
- 14 Traceability Matrix 101: Why It’s Not the Ultimate Solution for Managing Requirements
- 15 Live Traceability vs. After-the-Fact Traceability
- 16 Overcoming Barriers to Live Requirements Traceability™
- 17 Requirements Traceability, What Are You Missing?
- 18 Four Best Practices for Requirements Traceability
- 19 Requirements Traceability: Links in the Chain
- 20 What Are the Benefits of End-to-End Traceability During Product Development?
- 21 FAQs About Requirements Traceability
- 5. Requirements Management Tools and Software
- Overview
- 1 Selecting the Right Requirements Management Tools and Software
- 2 Why Investing in Requirements Management Software Makes Business Sense During an Economic Downturn
- 3 Why Word and Excel Alone is Not Enough for Product, Software, and Systems Development
- 4 Can You Track Requirements in Excel?
- 5 What Is Application Lifecycle Management (ALM)?
- 6 Is There Life After DOORS®?
- 7 Can You Track Requirements in Jira?
- 8 Checklist: Selecting a Requirements Management Tool
- 6. Requirements Validation and Verification
- 7. Meeting Regulatory Compliance and Industry Standards
- Overview
- 1 Understanding ISO Standards
- 2 Understanding ISO/IEC 27001: A Guide to Information Security Management
- 3 What is DevSecOps? A Guide to Building Secure Software
- 4 Compliance Management
- 5 What is FMEA? Failure Mode and Effects Analysis Guide
- 6 TÜV SÜD: Ensuring Safety, Quality, and Sustainability Worldwide
- 8. Systems Engineering
- Overview
- 1 What is Systems Engineering?
- 2 How Do Engineers Collaborate? A Guide to Streamlined Teamwork and Innovation
- 3 The Systems Engineering Body of Knowledge (SEBoK)
- 4 What is MBSE? Model-Based Systems Engineering Explained
- 5 Digital Engineering Between Government and Contractors
- 6 Digital Engineering Tools: The Key to Driving Innovation and Efficiency in Complex Systems
- 9. Automotive Development
- 10. Medical Device & Life Sciences Development
- Overview
- 1 The Importance of Benefit-Risk Analysis in Medical Device Development
- 2 Software as a Medical Device: Revolutionizing Healthcare
- 3 What’s a Design History File, and How Are DHFs Used by Product Teams?
- 4 Navigating the Risks of Software of Unknown Pedigree (SOUP) in the Medical Device & Life Sciences Industry
- 5 What is ISO 13485? Your Comprehensive Guide to Compliant Medical Device Manufacturing
- 6 What You Need to Know: ANSI/AAMI SW96:2023 — Medical Device Security
- 7 ISO 13485 vs ISO 9001: Understanding the Differences and Synergies
- 8 What Is IEC 62304? A Guide to Medical Device Software
- 9 What Is a Device Master Record (DMR)? Definition and FDA Requirements
- 10 Failure Modes, Effects, and Diagnostic Analysis (FMEDA) for Medical Devices: What You Need to Know
- 11 Embracing the Future of Healthcare: Exploring the Internet of Medical Things (IoMT)
- 12 What Is General Safety and Performance Requirements (GSPR)? What You Need To Know
- 11. Aerospace & Defense Development
- 12. Architecture, Engineering, and Construction (AEC industry) Development
- 13. Industrial Manufacturing & Machinery, Automation & Robotics, Consumer Electronics, and Energy
- 14. Semiconductor Development
- 15. AI in Product Development
- 16. Risk Management
- 17. Product Development Terms and Definitions
Chapter 1: What is Requirements Management? A Complete Guide
Chapters
- 1. Requirements Management
- Overview
- 1 What is Requirements Management? A Complete Guide
- 2 Why do you need Requirements Management?
- 3 Four Stages of Requirements Management Processes
- 4 Adopting an Agile Approach to Requirements Management
- 5 Status Request Changes
- 6 Conquering the 5 Biggest Challenges of Requirements Management
- 7 Three Reasons You Need a Requirements Management Solution
- 8 Guide to Poor Requirements: Identify Causes, Repercussions, and How to Fix Them
- 2. Writing Requirements
- Overview
- 1 Functional requirements examples and templates
- 2 What Is a Product Requirements Document? A Complete PRD Guide
- 3 Identifying and Measuring Requirements Quality
- 4 What Is a User Requirement Specification (URS)? How to Write and Manage One
- 5 How to Write a System Requirements Specification (SRS) Document
- 6 The Fundamentals of Business Requirements: Examples of Business Requirements and the Importance of Excellence
- 7 Adopting the EARS Notation to Improve Requirements Engineering
- 8 What Is a Compliance Risk Assessment? Steps, Framework, and Examples
- 9 Jama Connect Advisor™
- 10 Frequently Asked Questions about the EARS Notation and Jama Connect Advisor™
- 11 How to Write an Effective Product Requirements Document (PRD)
- 12 Functional vs. Non-Functional Requirements
- 13 What Are Nonfunctional Requirements and How Do They Impact Product Development?
- 14 Characteristics of Effective Software Requirements and Software Requirements Specifications (SRS)
- 15 What Is a Software Design Specification? Key Components + Template
- 16 8 Do’s and Don’ts for Writing Requirements
- 3. Requirements Gathering and Management Processes
- Overview
- 1 Requirements Engineering
- 2 Requirements Analysis
- 3 A Guide to Requirements Elicitation for Product Teams
- 4 Requirements Gathering Techniques for Agile Product Teams
- 5 Requirements Gathering in Software Engineering: Process, Techniques, and Best Practices
- 6 Defining and Implementing a Requirements Baseline
- 7 Managing Project Scope — Why It Matters and Best Practices
- 8 How Long Do Requirements Take?
- 9 How to Reuse Requirements Across Multiple Products
- 4. Requirements Traceability
- Overview
- 1 How is Traceability Achieved? A Practical Guide for Engineers
- 2 What is Requirements Traceability? Importance Explained
- 3 Tracing Your Way to Success: The Crucial Role of Traceability in Modern Product and Systems Development
- 4 Bidirectional Traceability: What It Is and How to Implement It
- 5 What is Engineering Change Management (ECM)? A Complete Guide
- 6 Change Impact Analysis (CIA): A Short Guide for Effective Implementation
- 7 What is Meant by Version Control?
- 8 What is Requirements Traceability and Why Does It Matter for Product Teams?
- 9 Key Traceability Challenges and Tips for Ensuring Accountability and Efficiency
- 10 The Role of a Data Thread in Product and Software Development
- 11 Unraveling the Digital Thread: Enhancing Connectivity and Efficiency
- 12 What is a Traceability Matrix? A Guide to Requirements Traceability
- 13 How to Create and Use a Requirements Traceability Matrix (RTM)
- 14 Traceability Matrix 101: Why It’s Not the Ultimate Solution for Managing Requirements
- 15 Live Traceability vs. After-the-Fact Traceability
- 16 Overcoming Barriers to Live Requirements Traceability™
- 17 Requirements Traceability, What Are You Missing?
- 18 Four Best Practices for Requirements Traceability
- 19 Requirements Traceability: Links in the Chain
- 20 What Are the Benefits of End-to-End Traceability During Product Development?
- 21 FAQs About Requirements Traceability
- 5. Requirements Management Tools and Software
- Overview
- 1 Selecting the Right Requirements Management Tools and Software
- 2 Why Investing in Requirements Management Software Makes Business Sense During an Economic Downturn
- 3 Why Word and Excel Alone is Not Enough for Product, Software, and Systems Development
- 4 Can You Track Requirements in Excel?
- 5 What Is Application Lifecycle Management (ALM)?
- 6 Is There Life After DOORS®?
- 7 Can You Track Requirements in Jira?
- 8 Checklist: Selecting a Requirements Management Tool
- 6. Requirements Validation and Verification
- 7. Meeting Regulatory Compliance and Industry Standards
- Overview
- 1 Understanding ISO Standards
- 2 Understanding ISO/IEC 27001: A Guide to Information Security Management
- 3 What is DevSecOps? A Guide to Building Secure Software
- 4 Compliance Management
- 5 What is FMEA? Failure Mode and Effects Analysis Guide
- 6 TÜV SÜD: Ensuring Safety, Quality, and Sustainability Worldwide
- 8. Systems Engineering
- Overview
- 1 What is Systems Engineering?
- 2 How Do Engineers Collaborate? A Guide to Streamlined Teamwork and Innovation
- 3 The Systems Engineering Body of Knowledge (SEBoK)
- 4 What is MBSE? Model-Based Systems Engineering Explained
- 5 Digital Engineering Between Government and Contractors
- 6 Digital Engineering Tools: The Key to Driving Innovation and Efficiency in Complex Systems
- 9. Automotive Development
- 10. Medical Device & Life Sciences Development
- Overview
- 1 The Importance of Benefit-Risk Analysis in Medical Device Development
- 2 Software as a Medical Device: Revolutionizing Healthcare
- 3 What’s a Design History File, and How Are DHFs Used by Product Teams?
- 4 Navigating the Risks of Software of Unknown Pedigree (SOUP) in the Medical Device & Life Sciences Industry
- 5 What is ISO 13485? Your Comprehensive Guide to Compliant Medical Device Manufacturing
- 6 What You Need to Know: ANSI/AAMI SW96:2023 — Medical Device Security
- 7 ISO 13485 vs ISO 9001: Understanding the Differences and Synergies
- 8 What Is IEC 62304? A Guide to Medical Device Software
- 9 What Is a Device Master Record (DMR)? Definition and FDA Requirements
- 10 Failure Modes, Effects, and Diagnostic Analysis (FMEDA) for Medical Devices: What You Need to Know
- 11 Embracing the Future of Healthcare: Exploring the Internet of Medical Things (IoMT)
- 12 What Is General Safety and Performance Requirements (GSPR)? What You Need To Know
- 11. Aerospace & Defense Development
- 12. Architecture, Engineering, and Construction (AEC industry) Development
- 13. Industrial Manufacturing & Machinery, Automation & Robotics, Consumer Electronics, and Energy
- 14. Semiconductor Development
- 15. AI in Product Development
- 16. Risk Management
- 17. Product Development Terms and Definitions
What is Requirements Management? A Complete Guide
A product team can have strong engineers and a solid timeline and still end up in rework if nobody defined what the product needed to do. Requirements management helps teams avoid that outcome. It means defining, tracking, and proving what a system must do across its lifecycle. And for teams building complex, regulated products, it’s one of the few things that directly affects both product quality and time to market.
Here’s how the process works, where teams get it wrong, and what effective requirements management looks like in regulated development.
What is Requirements Management?
Requirements management covers how teams identify, document, and track requirements throughout the lifecycle of a system, product, or service. A “need” is what someone expects the product to do, and a “requirement” is the formal, testable version of that need. ISO/IEC/IEEE 29148 frames this as iterative work that recurs throughout the lifecycle, not a one-time handoff.
Requirements Management vs. Project Management
Requirements management defines what the system must do and how well it must perform, while project management defines the work required to deliver it on time and within budget. The project manager tracks whether requirements work is on schedule, while the systems engineer tracks whether the requirements themselves are correct, complete, and traceable.
RELATED ARTICLE: Evaluating Requirements Management Tools
Why Requirements Management is Important
More than half of project defects trace back to requirements, and the majority of rework effort goes toward fixing them. Those failures tend to show up late, when the cost of change is highest, and they hit teams in a few predictable ways:
- Rework and cost overruns: Projects that invest 8 to 14% of total cost on requirements management see less than 60% cost overrun, while projects that invest less than 5% face 80 to 200% overruns.
- Cross-team alignment: A shared requirements baseline (a formally approved, locked version of all requirements) gives every team a clear view of what the system must do, who owns each requirement, and what depends on it. Without that, vague requirements like “the device shall respond quickly” can produce incompatible implementations that nobody catches until integration.
- Regulatory compliance: FDA 21 CFR 820.30 requires design inputs to be documented, reviewed, and approved. Standards like DO-178C (aerospace software safety) and ISO 26262 (automotive functional safety) expect bidirectional traceability across requirements, implementation, and verification. In medical devices, the FDA’s QMSR rule (in effect since February 2, 2026) raises these expectations further.
Without trace evidence, organizations risk delayed approvals, failed audits, or blocked market access. These problems get worse as projects grow, especially when teams aren’t precise about what kind of requirement they’re dealing with.
Ready to Find Out More?
Our team of experts is here to answer any questions and learn how we can help enable your continued success. Get started with a free 30-day trial, or book a demo!
Types of Requirements
Requirements are organized in layers, from why the product exists down to what it must do and how well.
| Business | Functional | Non-Functional | |
| Asks | Why does this product exist? | What must the system do? | How well must it perform? |
| Focus | Outcomes and goals | Measurable behaviors | Qualities and constraints |
| AEB example | Reduce pedestrian fatalities through collision avoidance | Brake within 200ms of detecting a pedestrian | Meet ASIL D safety and operate in rain, fog, low light |
| Written by | Product or business stakeholders | Systems engineers | Systems and domain engineers |
Business Requirements
Business requirements address the “why” behind a product and stay design-agnostic. For an autonomous emergency braking (AEB) system, a business requirement might read, “Reduce pedestrian fatalities in urban driving by providing automatic collision avoidance.” That defines the outcome without prescribing how the system should work.
Functional Requirements
Functional requirements translate business needs into measurable behaviors. For the same AEB system, a functional requirement might state, “The system shall initiate full braking force within 200 milliseconds of detecting a pedestrian in the vehicle’s forward path.” That’s testable, unambiguous, and traceable back to the business requirement above.
Non-Functional Requirements
Non-functional requirements define how well the system performs rather than what it does, covering qualities like reliability, safety, and maintainability. Continuing the AEB example, a non-functional requirement might state, “The system shall meet ISO 26262 ASIL D functional safety requirements and operate reliably at speeds between 10 and 60 km/h in rain, fog, and low-light conditions.”
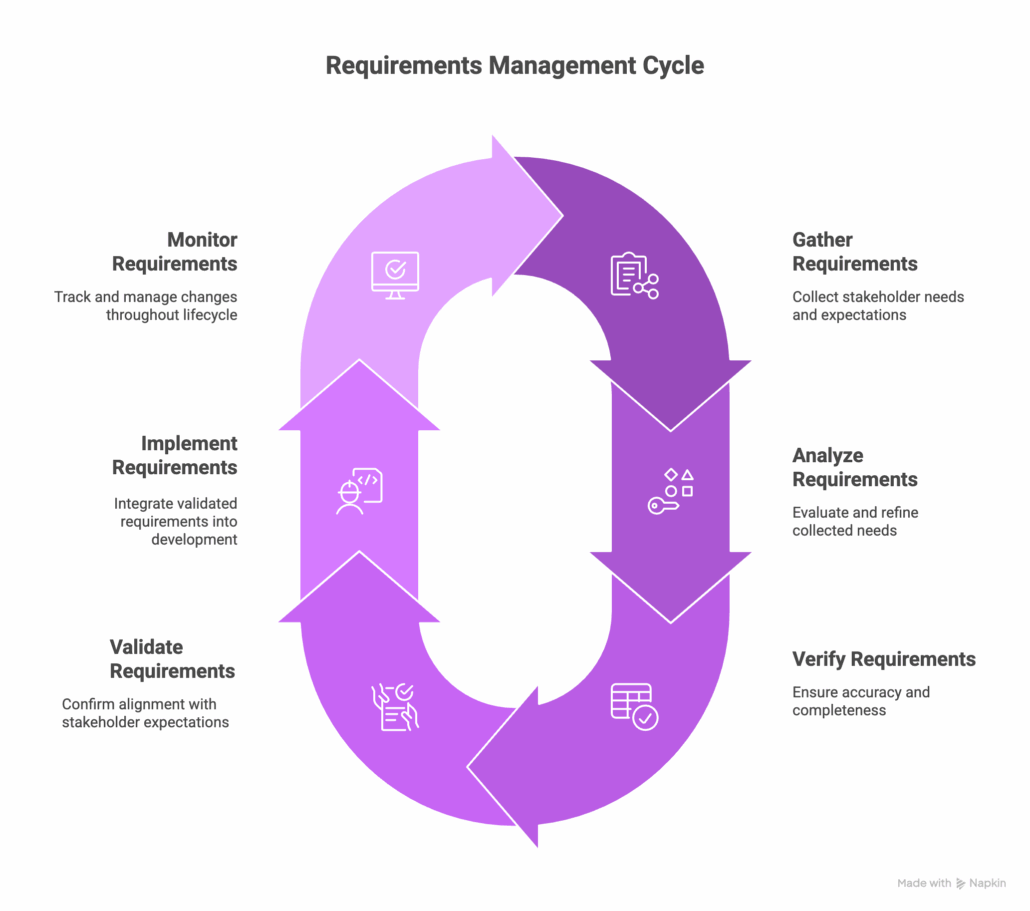
The Four Stages of Requirements Management
The requirements management process is often mapped to the V-model, though in practice it’s less of a strict sequence and more of a map showing how stages relate to each other. Teams move back and forth as their understanding of the product matures.
1. Requirements Elicitation
Elicitation captures raw inputs like stakeholder needs, regulatory constraints, use cases, and hazard analyses. One common mistake at this stage is recording a preferred solution as a need, which limits options when tradeoffs come up later.
2. Requirements Definition and Documentation
Once elicited, requirements need to be written in a verifiable format. INCOSE (the International Council on Systems Engineering) teaches the “shall” statement structure, where a subject, active verb, object, and qualifier form a testable statement. For example, “The autonomous taxi shall permit the passenger doors to open upon arriving at the destination.”
3. System Verification
Verification asks whether the team built the system right. Does the implementation match the specification? Most teams verify through testing, analysis, demonstration, or inspection. If any of those methods reveal a gap, the requirement or the implementation needs to be reconciled before moving forward.
4. System Validation
Validation asks the opposite: whether the team built the right system. When validation fails, the root cause is usually a missing need or a bad assumption from elicitation, which is why teams circle back to earlier stages even late in development.
Requirements Traceability
Teams also need a way to track how each requirement connects to the designs, tests, and risk controls built around it. Requirements traceability is the ability to follow a requirement in both forward and backward directions, from origin through deployment and refinement. Without traceability, teams struggle to confirm coverage, assess change impact, or produce the audit artifacts regulated programs require.
What is a Requirements Traceability Matrix (RTM)?
A requirements traceability matrix (RTM) maps how stakeholder needs, system requirements, design elements, and verification activities connect to each other. Each row links a requirement to its parent need, the design elements that address it, the test cases that verify it, and the results of those tests.
Teams generate RTMs for audits and milestones, but the real day-to-day value is knowing what’s affected when something changes. If a stakeholder need shifts, the RTM shows exactly which requirements, designs, and tests need to be reviewed.
Building Bidirectional Traceability
Bidirectional traceability means every requirement traces forward to its implementation and verification, and every test case traces backward to the requirement it satisfies. Without the backward link, passing tests don’t actually prove the right requirement was verified.
How to Manage Requirements Changes
Traceability shows what’s connected. Change control keeps those connections accurate. After baseline, every change needs a controlled process. Without it, teams ship mismatched versions of requirements, tests, and risk controls. In practice, that control breaks down into two areas:
- Change control workflows: A typical workflow moves through change request initiation, routing, impact assessment, and formal approval with electronic signatures. The workflow matters less than the evidence trail it leaves, since auditors want to see who approved a change, when, and what was reviewed.
- Impact analysis and version control: A change to one requirement can ripple through downstream test cases, risk assessments, and supplier interfaces. Effective traceability makes that chain visible before the change is approved. For example, if a braking-distance requirement changes from 40 meters to 35 meters, that change affects the linked hazard analysis, the FMEA (Failure Mode and Effects Analysis) severity rating for brake failure, and every test case that validated the original threshold. Formal baselines create fixed snapshots at key milestones and support both engineering decisions and regulatory evidence.
Without these controls, small changes pile up into mismatched artifacts that only show up during integration or audit.
Common Requirements Management Challenges
Requirements problems at scale rarely come from one bad “shall” statement. They grow out of volume and coupling, where too many stakeholders, interfaces, and dependencies overwhelm the tools teams are using to manage them:
- Last-minute feedback and decision rehashing: When stakeholders can’t review requirements in a structured way, feedback arrives late and reopens decisions that downstream teams already acted on. Purpose-built tools address this with formal review workflows that assign roles, collect signatures, and track status.
- The hidden cost of change: Every modification to a requirement triggers updates across connected artifacts, and tracking those changes in Word documents creates a time tax that compounds as the product grows. It shows up as missed links, mismatched versions, and test suites that no longer match current requirements.
- Document-centric approaches breaking down: Word and Excel work for small teams with a handful of requirements and no regulatory pressure. They break down when nobody can maintain traceability across thousands of requirements, hundreds of test cases, and dozens of risk items.
Moving to a dedicated tool solves the traceability and version control problems, but the tool only works if the underlying requirements practices are solid.
Requirements Management Best Practices
The teams that handle requirements management well as projects grow tend to do three things consistently.
Write Clear, Testable Requirements
INCOSE describes well-written requirements as unambiguous, complete, feasible, verifiable, and conforming. The EARS (Easy Approach to Requirements Syntax) methodology is one way to enforce clarity, especially for requirements that read like intent statements but don’t map to a verification method:
- Ubiquitous pattern: “The mobile phone shall have a mass of less than XX grams.” This applies when the requirement is always active.
- Event-driven pattern: “When the user selects caller count, the software shall display a count of participants.” This applies when a trigger initiates the response.
Explicit triggers and responses leave less room for argument during verification planning.
Engage Stakeholders Early and Often
INCOSE recommends planning who you’ll talk to, what you need from them, and where the handoffs between teams happen. That prevents the common problem where interface requirements only surface after subsystem teams have already started design.
Structure Requirements for Reusability
Product line engineering requires reusable requirements. INCOSE describes approaches including cloning, referencing with a maintained link, and modular configuration. Purpose-built tools can maintain live links between source and derivative requirements, making it easier to track differences and propagate updates.
How to Choose a Requirements Management Tool
When you pick a tool matters as much as what’s on the feature list. Traceability complexity grows fast, and migrations introduce gaps when teams switch after baselining.
Features to Look For
In regulated development, a requirements management tool needs to produce clean proof of what you did and why, so you should look for these capabilities:
- Bidirectional traceability: End-to-end trace from stakeholder needs through test results, with automated orphan detection (flagging requirements with no linked test or design element).
- Compliance workflows: Electronic signatures, approval routing, and auditable history.
- Integration: Sync with Jira, Azure DevOps, ReqIF (a standard interchange format for requirements data), and an open API.
- Change control: Formal baselines, comparison, and impact analysis across relationships.
- Scalability: The platform should handle growing requirements volume and team size without degrading performance. Enterprise programs add thousands of requirements over time, and a tool that slows down or forces workarounds at scale creates the same gaps it was meant to prevent.
When a tool is weak in any of these areas, the work shifts into spreadsheets and traceability gaps pile up.
Integration with Existing Workflows
INCOSE warns against selecting a tool in isolation or prioritizing lowest cost over fit. Teams running Agile alongside formal baselines should look for bidirectional sync with tools like Jira so developers can keep working in their preferred environment while the trace chain remains intact.
Getting Requirements Management Right
The biggest risk in requirements management is quiet drift, where small gaps between requirements, tests, and risk controls build up over time and show up during integration, audits, or late-stage validation. The teams that avoid this invest in traceability early, enforce change control at baseline, and use tooling designed for that level of complexity.
Jama Connect®, a requirements management platform for regulated product development, supports this workflow. When a requirement changes, Live Traceability™ automatically identifies each linked design element, test case, and risk control. Traceability Information Models (TIMs) define expected relationships so missing links surface early for review. The platform is also built to scale with enterprise programs as requirements volume and team size grow. Jama Connect Interchange™ provides bidirectional sync with Jira, so Agile teams stay connected to the trace chain without leaving their preferred workflow.
Start your free 30-day trial to see how Jama Connect connects requirements to designs, tests, and risk controls in one place.
Frequently Asked Questions About Requirements Management
What is a requirements management plan?
A requirements management plan defines how an organization will identify, document, trace, and control requirements throughout the product lifecycle. It covers roles, tools, approval workflows, and how changes get evaluated.
Who owns the requirements management process?
Ownership is usually shared. Systems engineering owns definition and verification planning, while quality and regulatory teams own governance, review rules, and audit readiness.
What is the difference between verification and validation?
Verification checks whether the system was built right, meaning the implementation matches the specification. Validation checks whether the right system was built, meaning the product actually does what users need in its real-world environment.
How does requirements management reduce project risk?
It catches ambiguity before it becomes a design conflict and controls change so teams aren’t building to outdated baselines. In regulated programs, it also reduces audit risk by providing clear traceability between requirements, implementation, and verification.
RELATED ARTICLE: Evaluating Requirements Management Tools
In This Webinar, Learn More About Standardizing Requirements Management Across the Organization
Requirements Management is the process of gathering, analyzing, verifying, and validating the needs and requirements for the given product or system being developed. Successful requirements management ensures that completed deliverables meet the expectations of the stakeholders.
Book a Demo
See Jama Connect in Action!
Our Jama Connect experts are ready to guide you through a personalized demo, answer your questions, and show you how Jama Connect can help you identify risks, improve cross-team collaboration, and drive faster time to market.
