Bidirectional Traceability: What It Is and How to Implement It
The Essential Guide to Requirements Management and Traceability
Chapters
- 1. Requirements Management
- Overview
- 1 What is Requirements Management? A Complete Guide
- 2 Why do you need Requirements Management?
- 3 Four Stages of Requirements Management Processes
- 4 Adopting an Agile Approach to Requirements Management
- 5 Status Request Changes
- 6 Conquering the 5 Biggest Challenges of Requirements Management
- 7 Three Reasons You Need a Requirements Management Solution
- 8 Guide to Poor Requirements: Identify Causes, Repercussions, and How to Fix Them
- 2. Writing Requirements
- Overview
- 1 Functional requirements examples and templates
- 2 Identifying and Measuring Requirements Quality
- 3 How to Write a System Requirements Specification (SRS) Document
- 4 The Fundamentals of Business Requirements: Examples of Business Requirements and the Importance of Excellence
- 5 Adopting the EARS Notation to Improve Requirements Engineering
- 6 Jama Connect Advisor™
- 7 Frequently Asked Questions about the EARS Notation and Jama Connect Advisor™
- 8 How to Write an Effective Product Requirements Document (PRD)
- 9 Functional vs. Non-Functional Requirements
- 10 What Are Nonfunctional Requirements and How Do They Impact Product Development?
- 11 Characteristics of Effective Software Requirements and Software Requirements Specifications (SRS)
- 12 8 Do’s and Don’ts for Writing Requirements
- 3. Requirements Gathering and Management Processes
- Overview
- 1 Requirements Engineering
- 2 Requirements Analysis
- 3 A Guide to Requirements Elicitation for Product Teams
- 4 Requirements Gathering Techniques for Agile Product Teams
- 5 What is Requirements Gathering in Software Engineering?
- 6 Defining and Implementing a Requirements Baseline
- 7 Managing Project Scope — Why It Matters and Best Practices
- 8 How Long Do Requirements Take?
- 9 How to Reuse Requirements Across Multiple Products
- 4. Requirements Traceability
- Overview
- 1 How is Traceability Achieved? A Practical Guide for Engineers
- 2 What is Requirements Traceability? Importance Explained
- 3 Tracing Your Way to Success: The Crucial Role of Traceability in Modern Product and Systems Development
- 4 Bidirectional Traceability: What It Is and How to Implement It
- 5 What is Engineering Change Management (ECM)? A Complete Guide
- 6 Change Impact Analysis (CIA): A Short Guide for Effective Implementation
- 7 What is Meant by Version Control?
- 8 What is Requirements Traceability and Why Does It Matter for Product Teams?
- 9 Key Traceability Challenges and Tips for Ensuring Accountability and Efficiency
- 10 The Role of a Data Thread in Product and Software Development
- 11 Unraveling the Digital Thread: Enhancing Connectivity and Efficiency
- 12 What is a Traceability Matrix? A Guide to Requirements Traceability
- 13 How to Create and Use a Requirements Traceability Matrix (RTM)
- 14 Traceability Matrix 101: Why It’s Not the Ultimate Solution for Managing Requirements
- 15 Live Traceability vs. After-the-Fact Traceability
- 16 Overcoming Barriers to Live Requirements Traceability™
- 17 Requirements Traceability, What Are You Missing?
- 18 Four Best Practices for Requirements Traceability
- 19 Requirements Traceability: Links in the Chain
- 20 What Are the Benefits of End-to-End Traceability During Product Development?
- 21 FAQs About Requirements Traceability
- 5. Requirements Management Tools and Software
- Overview
- 1 Selecting the Right Requirements Management Tools and Software
- 2 Why Investing in Requirements Management Software Makes Business Sense During an Economic Downturn
- 3 Why Word and Excel Alone is Not Enough for Product, Software, and Systems Development
- 4 Can You Track Requirements in Excel?
- 5 Application lifecycle management (ALM)
- 6 Is There Life After DOORS®?
- 7 Can You Track Requirements in Jira?
- 8 Checklist: Selecting a Requirements Management Tool
- 6. Requirements Validation and Verification
- 7. Meeting Regulatory Compliance and Industry Standards
- Overview
- 1 Understanding ISO Standards
- 2 Understanding ISO/IEC 27001: A Guide to Information Security Management
- 3 What is DevSecOps? A Guide to Building Secure Software
- 4 Compliance Management
- 5 What is FMEA? Failure Mode and Effects Analysis Guide
- 6 TÜV SÜD: Ensuring Safety, Quality, and Sustainability Worldwide
- 8. Systems Engineering
- Overview
- 1 What is Systems Engineering?
- 2 How Do Engineers Collaborate? A Guide to Streamlined Teamwork and Innovation
- 3 The Systems Engineering Body of Knowledge (SEBoK)
- 4 What is MBSE? Model-Based Systems Engineering Explained
- 5 Digital Engineering Between Government and Contractors
- 6 Digital Engineering Tools: The Key to Driving Innovation and Efficiency in Complex Systems
- 9. Automotive Development
- 10. Medical Device & Life Sciences Development
- Overview
- 1 The Importance of Benefit-Risk Analysis in Medical Device Development
- 2 Software as a Medical Device: Revolutionizing Healthcare
- 3 What’s a Design History File, and How Are DHFs Used by Product Teams?
- 4 Navigating the Risks of Software of Unknown Pedigree (SOUP) in the Medical Device & Life Sciences Industry
- 5 What is ISO 13485? Your Comprehensive Guide to Compliant Medical Device Manufacturing
- 6 What You Need to Know: ANSI/AAMI SW96:2023 — Medical Device Security
- 7 ISO 13485 vs ISO 9001: Understanding the Differences and Synergies
- 8 Failure Modes, Effects, and Diagnostic Analysis (FMEDA) for Medical Devices: What You Need to Know
- 9 Embracing the Future of Healthcare: Exploring the Internet of Medical Things (IoMT)
- 11. Aerospace & Defense Development
- 12. Architecture, Engineering, and Construction (AEC industry) Development
- 13. Industrial Manufacturing & Machinery, Automation & Robotics, Consumer Electronics, and Energy
- 14. Semiconductor Development
- 15. AI in Product Development
- 16. Risk Management
- 17. Product Development Terms and Definitions
Chapter 4: Bidirectional Traceability: What It Is and How to Implement It
Chapters
- 1. Requirements Management
- Overview
- 1 What is Requirements Management? A Complete Guide
- 2 Why do you need Requirements Management?
- 3 Four Stages of Requirements Management Processes
- 4 Adopting an Agile Approach to Requirements Management
- 5 Status Request Changes
- 6 Conquering the 5 Biggest Challenges of Requirements Management
- 7 Three Reasons You Need a Requirements Management Solution
- 8 Guide to Poor Requirements: Identify Causes, Repercussions, and How to Fix Them
- 2. Writing Requirements
- Overview
- 1 Functional requirements examples and templates
- 2 Identifying and Measuring Requirements Quality
- 3 How to Write a System Requirements Specification (SRS) Document
- 4 The Fundamentals of Business Requirements: Examples of Business Requirements and the Importance of Excellence
- 5 Adopting the EARS Notation to Improve Requirements Engineering
- 6 Jama Connect Advisor™
- 7 Frequently Asked Questions about the EARS Notation and Jama Connect Advisor™
- 8 How to Write an Effective Product Requirements Document (PRD)
- 9 Functional vs. Non-Functional Requirements
- 10 What Are Nonfunctional Requirements and How Do They Impact Product Development?
- 11 Characteristics of Effective Software Requirements and Software Requirements Specifications (SRS)
- 12 8 Do’s and Don’ts for Writing Requirements
- 3. Requirements Gathering and Management Processes
- Overview
- 1 Requirements Engineering
- 2 Requirements Analysis
- 3 A Guide to Requirements Elicitation for Product Teams
- 4 Requirements Gathering Techniques for Agile Product Teams
- 5 What is Requirements Gathering in Software Engineering?
- 6 Defining and Implementing a Requirements Baseline
- 7 Managing Project Scope — Why It Matters and Best Practices
- 8 How Long Do Requirements Take?
- 9 How to Reuse Requirements Across Multiple Products
- 4. Requirements Traceability
- Overview
- 1 How is Traceability Achieved? A Practical Guide for Engineers
- 2 What is Requirements Traceability? Importance Explained
- 3 Tracing Your Way to Success: The Crucial Role of Traceability in Modern Product and Systems Development
- 4 Bidirectional Traceability: What It Is and How to Implement It
- 5 What is Engineering Change Management (ECM)? A Complete Guide
- 6 Change Impact Analysis (CIA): A Short Guide for Effective Implementation
- 7 What is Meant by Version Control?
- 8 What is Requirements Traceability and Why Does It Matter for Product Teams?
- 9 Key Traceability Challenges and Tips for Ensuring Accountability and Efficiency
- 10 The Role of a Data Thread in Product and Software Development
- 11 Unraveling the Digital Thread: Enhancing Connectivity and Efficiency
- 12 What is a Traceability Matrix? A Guide to Requirements Traceability
- 13 How to Create and Use a Requirements Traceability Matrix (RTM)
- 14 Traceability Matrix 101: Why It’s Not the Ultimate Solution for Managing Requirements
- 15 Live Traceability vs. After-the-Fact Traceability
- 16 Overcoming Barriers to Live Requirements Traceability™
- 17 Requirements Traceability, What Are You Missing?
- 18 Four Best Practices for Requirements Traceability
- 19 Requirements Traceability: Links in the Chain
- 20 What Are the Benefits of End-to-End Traceability During Product Development?
- 21 FAQs About Requirements Traceability
- 5. Requirements Management Tools and Software
- Overview
- 1 Selecting the Right Requirements Management Tools and Software
- 2 Why Investing in Requirements Management Software Makes Business Sense During an Economic Downturn
- 3 Why Word and Excel Alone is Not Enough for Product, Software, and Systems Development
- 4 Can You Track Requirements in Excel?
- 5 Application lifecycle management (ALM)
- 6 Is There Life After DOORS®?
- 7 Can You Track Requirements in Jira?
- 8 Checklist: Selecting a Requirements Management Tool
- 6. Requirements Validation and Verification
- 7. Meeting Regulatory Compliance and Industry Standards
- Overview
- 1 Understanding ISO Standards
- 2 Understanding ISO/IEC 27001: A Guide to Information Security Management
- 3 What is DevSecOps? A Guide to Building Secure Software
- 4 Compliance Management
- 5 What is FMEA? Failure Mode and Effects Analysis Guide
- 6 TÜV SÜD: Ensuring Safety, Quality, and Sustainability Worldwide
- 8. Systems Engineering
- Overview
- 1 What is Systems Engineering?
- 2 How Do Engineers Collaborate? A Guide to Streamlined Teamwork and Innovation
- 3 The Systems Engineering Body of Knowledge (SEBoK)
- 4 What is MBSE? Model-Based Systems Engineering Explained
- 5 Digital Engineering Between Government and Contractors
- 6 Digital Engineering Tools: The Key to Driving Innovation and Efficiency in Complex Systems
- 9. Automotive Development
- 10. Medical Device & Life Sciences Development
- Overview
- 1 The Importance of Benefit-Risk Analysis in Medical Device Development
- 2 Software as a Medical Device: Revolutionizing Healthcare
- 3 What’s a Design History File, and How Are DHFs Used by Product Teams?
- 4 Navigating the Risks of Software of Unknown Pedigree (SOUP) in the Medical Device & Life Sciences Industry
- 5 What is ISO 13485? Your Comprehensive Guide to Compliant Medical Device Manufacturing
- 6 What You Need to Know: ANSI/AAMI SW96:2023 — Medical Device Security
- 7 ISO 13485 vs ISO 9001: Understanding the Differences and Synergies
- 8 Failure Modes, Effects, and Diagnostic Analysis (FMEDA) for Medical Devices: What You Need to Know
- 9 Embracing the Future of Healthcare: Exploring the Internet of Medical Things (IoMT)
- 11. Aerospace & Defense Development
- 12. Architecture, Engineering, and Construction (AEC industry) Development
- 13. Industrial Manufacturing & Machinery, Automation & Robotics, Consumer Electronics, and Energy
- 14. Semiconductor Development
- 15. AI in Product Development
- 16. Risk Management
- 17. Product Development Terms and Definitions
Bidirectional Traceability: What It Is and How to Implement It
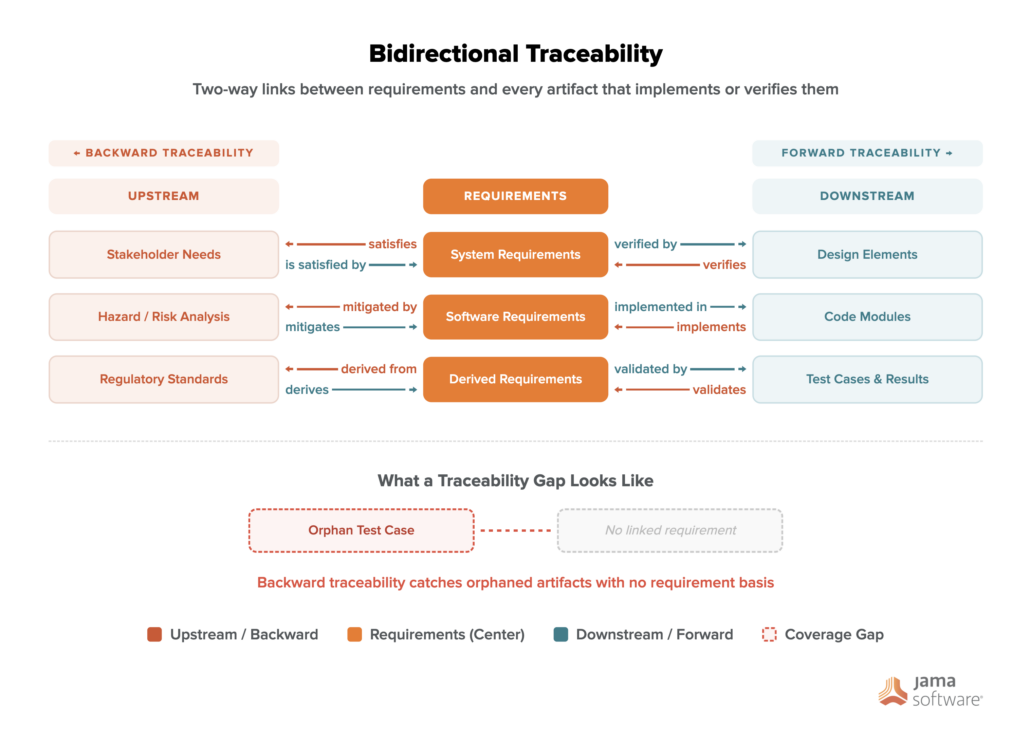
Bidirectional traceability gives engineering teams a two-way link between requirements and every artifact that implements or verifies them. Teams that maintain bidirectional traceability answer change impact questions on demand and walk into audits with coverage that’s already complete.
This guide covers what bidirectional traceability is, how it compares to forward and backward traceability, what safety standards require, and how to implement it.
What Is Bidirectional Traceability?
Bidirectional traceability is the ability to establish a two-way link between entities such that each has knowledge of the other. ISO 16404:2020 classifies it as a program management requirement because it supports both top-down impact analysis and bottom-up coverage analysis (also known as upstream and downstream links).
Although the relationship exists in both directions, it’s different in each direction. A system requirement “satisfies” a stakeholder need, while that stakeholder need “is satisfied by” the system requirement. Both links must be explicitly defined, stored, and maintained, because tools that treat them as interchangeable miss that distinction.
Why Bidirectional Traceability Matters for Product Development
In product development, rework often traces back to requirements problems. CMMI Level 2 formalizes this in Specific Practice 1.4, “Maintain Bidirectional Traceability of Requirements,” for good reason. When teams can prove full coverage in both directions, the results show up in three areas:
- Lower defect rates: Traceability completeness directly affects software quality. Fewer gaps in trace coverage mean fewer escaped defects.
- Faster change impact analysis: When a requirement changes, bidirectional links let you identify every affected work product immediately instead of reconstructing the impact by hand.
- Reduced rework at integration: Teams that catch coverage gaps and unintended scope early avoid the compounding cost of discovering them during system test or validation.
These gains depend on coverage in both directions, and each direction serves a different purpose.
RELATED ARTICLE: The Jama Software Guide to Requirements Traceability
Forward, Backward, and Bidirectional Traceability Compared
Each traceability direction answers a different question, and audits typically expect teams to demonstrate all three. The sections below break down how each direction works and where one-direction traceability falls short during change.
Forward Traceability
Forward traceability traces requirements downstream through the development lifecycle. It answers the question, “Have all requirements been implemented and verified?” For example, for an insulin infusion pump, forward traceability maps a user safety requirement like “prevent overdose” through hazard analysis, software safety requirements, safety mechanism design, and verification tests. Without it, teams can’t confirm that every requirement has corresponding verification evidence.
Forward traceability covers two distinct paths. The first traces from customer needs to requirements, confirming every need is captured. The second traces from requirements to test cases and design outputs, confirming each requirement is verified. Both paths apply to derived requirements as well (those not explicitly stated but necessary for the system to meet all defined ones).
Backward Traceability
Backward traceability works in the opposite direction. It traces from implementation artifacts and test results back to originating requirements and answers, “Does every work product tie to a requirement?” For the insulin pump example, that means every test case and every software module should connect back to a documented requirement. Code or tests that exist without a documented requirement to justify them, known as gold plating, have no place in a certified build.
Bidirectional Traceability: The Complete View
Bidirectional traceability combines both directions into a unified chain. The combined view makes change impact analysis practical. If the insulin pump’s “prevent overdose” requirement is revised, bidirectional traceability tells you exactly which tests, code modules, and design elements need to be reviewed before the next build.
Without both directions, teams can confirm that requirements have tests, but can’t confirm that every test traces to a current requirement, or vice versa. The two-way view closes both gaps at once.
Ready to Find Out More?
Our team of experts is here to answer any questions and learn how we can help enable your continued success. Get started with a free 30-day trial, or book a demo!
How Bidirectional Traceability Supports Regulatory Compliance
Safety-critical standards across aerospace, medical devices, and automotive share a common principle. Every requirement must connect to evidence of its fulfillment, and that connection must be navigable in both directions. Process frameworks like ISO/IEC/IEEE 29148 and CMMI Level 2 formalize this expectation as well. The differences across standards come down to scope, the artifacts each one expects, and how safety classification drives the depth of documentation.
Aerospace and Defense (DO-178C)
DO-178C requires verifiable trace data in both directions across system-level needs, software requirements, source code, test cases, and test results. Those links serve two purposes: confirming that every requirement has verification evidence, and ensuring that orphan code (implementation with no linked requirement) and dead code (code that can never execute) don’t slip into certified builds.
Medical Devices (IEC 62304)
IEC 62304 expects traceability across software requirements, risk management, and change control activities. Software requirements must be traceable to and from system requirements and risk analysis results. In practice, this requires bidirectional navigation across requirements, hazards, mitigations, and test documentation.
Automotive (ISO 26262)
Automotive functional safety programs aligned to ISO 26262 require traceable links between safety requirements, architecture decisions, implementation, and verification evidence. As safety integrity levels increase, verification expectations become more stringent. Many automotive organizations also typically use ASPICE process assessments, which examine whether teams can trace requirements and architecture into software work products and verification artifacts.
How to Implement Bidirectional Traceability
Bidirectional traceability runs on a requirements traceability matrix (RTM). The RTM records all trace links between requirements and the artifacts connected to them, including test cases, design elements, and code.
In a bidirectional RTM, requirements run down the center column with upstream items on the left and downstream verification items on the right, making coverage gaps visible in both directions at a glance.
Spreadsheet-based RTMs work at a small scale, but past a few hundred requirements, most systems engineering teams move to a dedicated requirements management tool. Once the RTM is in place, four practices keep bidirectional traceability working as requirements change:
Once the RTM is in place, four practices keep bidirectional traceability working as requirements change:
- Assign unique identifiers to every requirement: Requirements need stable, unique IDs with consistent metadata (owner, status, version, parent/child relationships) so links don’t break when items move between documents, tools, or teams.
- Define both link directions explicitly: For each link, capture both the forward and backward relationship types. A “satisfies” link and a “satisfied by” link carry different meanings and support different analyses.
- Validate coverage in both directions: Forward validation (requirements to tests) detects missing test coverage. Backward validation (tests to requirements) identifies orphaned tests with no requirement basis.
- Maintain traceability through changes: When a requirement changes, analyze the full impact before approving it, then propagate updates to all traced artifacts. Skipping this step is how test cases end up passing execution against requirements that have already changed.
Where teams run into trouble is consistent. The same three breakdown points appear across programs regardless of industry or team size.
Common Challenges With Bidirectional Traceability
Even teams that understand traceability well run into predictable failure modes when the process doesn’t support it. The most common problems fall into three categories.
Manual Maintenance in Spreadsheets
Spreadsheet-based RTMs break down once multiple people, versions, and baselines enter the picture. The failure modes are predictable as trace volume grows:
- Version drift: People edit local copies, then manually reconcile differences across email threads or shared drives.
- No suspect flagging: When an upstream requirement changes, nothing automatically marks downstream items as potentially out of date. Tools like Jama Connect® automate suspect flagging, but spreadsheets have no equivalent.
- No link semantics: Relationship meaning (satisfies vs. verifies) gets flattened into a single “X” cell.
- Audit fragility: A matrix can look complete while hiding stale links and outdated baselines.
Once upkeep becomes a dedicated weekly task, traceability stops being a control system and becomes a recurring documentation sprint.
Broken Links After Requirement Changes
When a requirement changes, all affected test cases, design elements, and risk items need to be identified and updated. Frequent requirement changes make this difficult even for experienced teams, especially when requirements aren’t tracked at a detailed level. The typical result is stale verification evidence that looks valid in the RTM but no longer matches the current requirement, which is exactly the kind of gap auditors catch.
Traceability Gaps Between Teams and Tools
Cross-team traceability breaks down when requirements, development, and verification live in different systems. Teams resort to manual workarounds like mapping IDs between tools or using code comments to reference specifications. The resulting blind spots tend to stay hidden until a reviewer or a failed integration exposes them. Integration platforms like Jama Connect Interchange™ address this by synchronizing trace data between requirements management tools and development environments like Jira.
RELATED ARTICLE: Achieving Live Traceability™ Using Jira®
Bidirectional Traceability Keeps Development Connected
Traceability problems in complex product development are rarely caused by teams that don’t understand traceability. They’re caused by processes that make maintaining it harder than it should be. When keeping trace links updated requires too much effort, gaps form silently and persist until something forces discovery. Requirements management tools change that dynamic by keeping trace data always current and flagging affected items automatically when an upstream requirement changes.
Jama Connect provides this through Live Traceability™ and Traceability Information Models (TIMs™), which detect when required downstream items are missing and flag affected artifacts automatically. Jama Connect Interchange™ extends that coverage to software teams in Jira, keeping both sides of the trace chain synchronized so gaps don’t form at the handoff. To see how it works firsthand, start a free 30-day trial today.
Frequently Asked Questions About Bidirectional Traceability
What is the difference between bidirectional and horizontal traceability?
Bidirectional describes the nature of the relationship (two-way vs. one-way). Horizontal describes the orientation across lifecycle artifacts at the same abstraction level, like linking requirements to design, risk assessments, and test cases. Vertical traceability moves up and down the architectural hierarchy. Bidirectional traceability can apply to both horizontal and vertical relationships.
Do agile teams need bidirectional traceability?
Yes, when they operate in regulated environments. Traceability between software requirements, system-level inputs, and risk analysis must work in both directions, regardless of development methodology. In practice, teams meet that expectation by linking backlog items to formal requirements and keeping two-way navigation intact between planning, implementation, and verification.
What standards require bidirectional traceability?
DO-178C requires two-way trace data across requirements, source code, and test evidence. ISO 26262 expects traceable links from safety goals through technical safety requirements to verification. IEC 62304 and FDA guidance require navigable connections between software requirements, risk analysis, and design outputs. CMMI Level 2 and ISO/IEC/IEEE 29148 also formalize the expectation.
What’s the fastest way to spot traceability gaps?
The fastest approach is to pull a report that shows parent requirements with no children and requirements with no linked verification items, then validate both directions. Check requirements to tests, and tests back to requirements. Once you can generate that view on demand, you can use the same checks for change impact analysis whenever a requirement revision comes up.
In This Webinar, We Cover Best Practices for Writing Requirements
Bidirectional Traceability: is the ability to establish a two-way link between entities such that each has knowledge of the other.
Book a Demo
See Jama Connect in Action!
Our Jama Connect experts are ready to guide you through a personalized demo, answer your questions, and show you how Jama Connect can help you identify risks, improve cross-team collaboration, and drive faster time to market.