What is a Traceability Matrix? A Guide to Requirements Traceability
The Essential Guide to Requirements Management and Traceability
Chapters
- 1. Requirements Management
- Overview
- 1 What is Requirements Management? A Complete Guide
- 2 Why do you need Requirements Management?
- 3 Four Stages of Requirements Management Processes
- 4 Adopting an Agile Approach to Requirements Management
- 5 Status Request Changes
- 6 Conquering the 5 Biggest Challenges of Requirements Management
- 7 Three Reasons You Need a Requirements Management Solution
- 8 Guide to Poor Requirements: Identify Causes, Repercussions, and How to Fix Them
- 2. Writing Requirements
- Overview
- 1 Functional requirements examples and templates
- 2 Identifying and Measuring Requirements Quality
- 3 How to Write a System Requirements Specification (SRS) Document
- 4 The Fundamentals of Business Requirements: Examples of Business Requirements and the Importance of Excellence
- 5 Adopting the EARS Notation to Improve Requirements Engineering
- 6 Jama Connect Advisor™
- 7 Frequently Asked Questions about the EARS Notation and Jama Connect Advisor™
- 8 How to Write an Effective Product Requirements Document (PRD)
- 9 Functional vs. Non-Functional Requirements
- 10 What Are Nonfunctional Requirements and How Do They Impact Product Development?
- 11 Characteristics of Effective Software Requirements and Software Requirements Specifications (SRS)
- 12 8 Do’s and Don’ts for Writing Requirements
- 3. Requirements Gathering and Management Processes
- Overview
- 1 Requirements Engineering
- 2 Requirements Analysis
- 3 A Guide to Requirements Elicitation for Product Teams
- 4 Requirements Gathering Techniques for Agile Product Teams
- 5 What is Requirements Gathering in Software Engineering?
- 6 Defining and Implementing a Requirements Baseline
- 7 Managing Project Scope — Why It Matters and Best Practices
- 8 How Long Do Requirements Take?
- 9 How to Reuse Requirements Across Multiple Products
- 4. Requirements Traceability
- Overview
- 1 How is Traceability Achieved? A Practical Guide for Engineers
- 2 What is Requirements Traceability? Importance Explained
- 3 Tracing Your Way to Success: The Crucial Role of Traceability in Modern Product and Systems Development
- 4 Bidirectional Traceability: What It Is and How to Implement It
- 5 What is Engineering Change Management (ECM)? A Complete Guide
- 6 Change Impact Analysis (CIA): A Short Guide for Effective Implementation
- 7 What is Meant by Version Control?
- 8 What is Requirements Traceability and Why Does It Matter for Product Teams?
- 9 Key Traceability Challenges and Tips for Ensuring Accountability and Efficiency
- 10 The Role of a Data Thread in Product and Software Development
- 11 Unraveling the Digital Thread: Enhancing Connectivity and Efficiency
- 12 What is a Traceability Matrix? A Guide to Requirements Traceability
- 13 How to Create and Use a Requirements Traceability Matrix (RTM)
- 14 Traceability Matrix 101: Why It’s Not the Ultimate Solution for Managing Requirements
- 15 Live Traceability vs. After-the-Fact Traceability
- 16 Overcoming Barriers to Live Requirements Traceability™
- 17 Requirements Traceability, What Are You Missing?
- 18 Four Best Practices for Requirements Traceability
- 19 Requirements Traceability: Links in the Chain
- 20 What Are the Benefits of End-to-End Traceability During Product Development?
- 21 FAQs About Requirements Traceability
- 5. Requirements Management Tools and Software
- Overview
- 1 Selecting the Right Requirements Management Tools and Software
- 2 Why Investing in Requirements Management Software Makes Business Sense During an Economic Downturn
- 3 Why Word and Excel Alone is Not Enough for Product, Software, and Systems Development
- 4 Can You Track Requirements in Excel?
- 5 Application lifecycle management (ALM)
- 6 Is There Life After DOORS®?
- 7 Can You Track Requirements in Jira?
- 8 Checklist: Selecting a Requirements Management Tool
- 6. Requirements Validation and Verification
- 7. Meeting Regulatory Compliance and Industry Standards
- Overview
- 1 Understanding ISO Standards
- 2 Understanding ISO/IEC 27001: A Guide to Information Security Management
- 3 What is DevSecOps? A Guide to Building Secure Software
- 4 Compliance Management
- 5 What is FMEA? Failure Mode and Effects Analysis Guide
- 6 TÜV SÜD: Ensuring Safety, Quality, and Sustainability Worldwide
- 8. Systems Engineering
- Overview
- 1 What is Systems Engineering?
- 2 How Do Engineers Collaborate? A Guide to Streamlined Teamwork and Innovation
- 3 The Systems Engineering Body of Knowledge (SEBoK)
- 4 What is MBSE? Model-Based Systems Engineering Explained
- 5 Digital Engineering Between Government and Contractors
- 6 Digital Engineering Tools: The Key to Driving Innovation and Efficiency in Complex Systems
- 9. Automotive Development
- 10. Medical Device & Life Sciences Development
- Overview
- 1 The Importance of Benefit-Risk Analysis in Medical Device Development
- 2 Software as a Medical Device: Revolutionizing Healthcare
- 3 What’s a Design History File, and How Are DHFs Used by Product Teams?
- 4 Navigating the Risks of Software of Unknown Pedigree (SOUP) in the Medical Device & Life Sciences Industry
- 5 What is ISO 13485? Your Comprehensive Guide to Compliant Medical Device Manufacturing
- 6 What You Need to Know: ANSI/AAMI SW96:2023 — Medical Device Security
- 7 ISO 13485 vs ISO 9001: Understanding the Differences and Synergies
- 8 Failure Modes, Effects, and Diagnostic Analysis (FMEDA) for Medical Devices: What You Need to Know
- 9 Embracing the Future of Healthcare: Exploring the Internet of Medical Things (IoMT)
- 11. Aerospace & Defense Development
- 12. Architecture, Engineering, and Construction (AEC industry) Development
- 13. Industrial Manufacturing & Machinery, Automation & Robotics, Consumer Electronics, and Energy
- 14. Semiconductor Development
- 15. AI in Product Development
- 16. Risk Management
- 17. Product Development Terms and Definitions
Chapter 4: What is a Traceability Matrix? A Guide to Requirements Traceability
Chapters
- 1. Requirements Management
- Overview
- 1 What is Requirements Management? A Complete Guide
- 2 Why do you need Requirements Management?
- 3 Four Stages of Requirements Management Processes
- 4 Adopting an Agile Approach to Requirements Management
- 5 Status Request Changes
- 6 Conquering the 5 Biggest Challenges of Requirements Management
- 7 Three Reasons You Need a Requirements Management Solution
- 8 Guide to Poor Requirements: Identify Causes, Repercussions, and How to Fix Them
- 2. Writing Requirements
- Overview
- 1 Functional requirements examples and templates
- 2 Identifying and Measuring Requirements Quality
- 3 How to Write a System Requirements Specification (SRS) Document
- 4 The Fundamentals of Business Requirements: Examples of Business Requirements and the Importance of Excellence
- 5 Adopting the EARS Notation to Improve Requirements Engineering
- 6 Jama Connect Advisor™
- 7 Frequently Asked Questions about the EARS Notation and Jama Connect Advisor™
- 8 How to Write an Effective Product Requirements Document (PRD)
- 9 Functional vs. Non-Functional Requirements
- 10 What Are Nonfunctional Requirements and How Do They Impact Product Development?
- 11 Characteristics of Effective Software Requirements and Software Requirements Specifications (SRS)
- 12 8 Do’s and Don’ts for Writing Requirements
- 3. Requirements Gathering and Management Processes
- Overview
- 1 Requirements Engineering
- 2 Requirements Analysis
- 3 A Guide to Requirements Elicitation for Product Teams
- 4 Requirements Gathering Techniques for Agile Product Teams
- 5 What is Requirements Gathering in Software Engineering?
- 6 Defining and Implementing a Requirements Baseline
- 7 Managing Project Scope — Why It Matters and Best Practices
- 8 How Long Do Requirements Take?
- 9 How to Reuse Requirements Across Multiple Products
- 4. Requirements Traceability
- Overview
- 1 How is Traceability Achieved? A Practical Guide for Engineers
- 2 What is Requirements Traceability? Importance Explained
- 3 Tracing Your Way to Success: The Crucial Role of Traceability in Modern Product and Systems Development
- 4 Bidirectional Traceability: What It Is and How to Implement It
- 5 What is Engineering Change Management (ECM)? A Complete Guide
- 6 Change Impact Analysis (CIA): A Short Guide for Effective Implementation
- 7 What is Meant by Version Control?
- 8 What is Requirements Traceability and Why Does It Matter for Product Teams?
- 9 Key Traceability Challenges and Tips for Ensuring Accountability and Efficiency
- 10 The Role of a Data Thread in Product and Software Development
- 11 Unraveling the Digital Thread: Enhancing Connectivity and Efficiency
- 12 What is a Traceability Matrix? A Guide to Requirements Traceability
- 13 How to Create and Use a Requirements Traceability Matrix (RTM)
- 14 Traceability Matrix 101: Why It’s Not the Ultimate Solution for Managing Requirements
- 15 Live Traceability vs. After-the-Fact Traceability
- 16 Overcoming Barriers to Live Requirements Traceability™
- 17 Requirements Traceability, What Are You Missing?
- 18 Four Best Practices for Requirements Traceability
- 19 Requirements Traceability: Links in the Chain
- 20 What Are the Benefits of End-to-End Traceability During Product Development?
- 21 FAQs About Requirements Traceability
- 5. Requirements Management Tools and Software
- Overview
- 1 Selecting the Right Requirements Management Tools and Software
- 2 Why Investing in Requirements Management Software Makes Business Sense During an Economic Downturn
- 3 Why Word and Excel Alone is Not Enough for Product, Software, and Systems Development
- 4 Can You Track Requirements in Excel?
- 5 Application lifecycle management (ALM)
- 6 Is There Life After DOORS®?
- 7 Can You Track Requirements in Jira?
- 8 Checklist: Selecting a Requirements Management Tool
- 6. Requirements Validation and Verification
- 7. Meeting Regulatory Compliance and Industry Standards
- Overview
- 1 Understanding ISO Standards
- 2 Understanding ISO/IEC 27001: A Guide to Information Security Management
- 3 What is DevSecOps? A Guide to Building Secure Software
- 4 Compliance Management
- 5 What is FMEA? Failure Mode and Effects Analysis Guide
- 6 TÜV SÜD: Ensuring Safety, Quality, and Sustainability Worldwide
- 8. Systems Engineering
- Overview
- 1 What is Systems Engineering?
- 2 How Do Engineers Collaborate? A Guide to Streamlined Teamwork and Innovation
- 3 The Systems Engineering Body of Knowledge (SEBoK)
- 4 What is MBSE? Model-Based Systems Engineering Explained
- 5 Digital Engineering Between Government and Contractors
- 6 Digital Engineering Tools: The Key to Driving Innovation and Efficiency in Complex Systems
- 9. Automotive Development
- 10. Medical Device & Life Sciences Development
- Overview
- 1 The Importance of Benefit-Risk Analysis in Medical Device Development
- 2 Software as a Medical Device: Revolutionizing Healthcare
- 3 What’s a Design History File, and How Are DHFs Used by Product Teams?
- 4 Navigating the Risks of Software of Unknown Pedigree (SOUP) in the Medical Device & Life Sciences Industry
- 5 What is ISO 13485? Your Comprehensive Guide to Compliant Medical Device Manufacturing
- 6 What You Need to Know: ANSI/AAMI SW96:2023 — Medical Device Security
- 7 ISO 13485 vs ISO 9001: Understanding the Differences and Synergies
- 8 Failure Modes, Effects, and Diagnostic Analysis (FMEDA) for Medical Devices: What You Need to Know
- 9 Embracing the Future of Healthcare: Exploring the Internet of Medical Things (IoMT)
- 11. Aerospace & Defense Development
- 12. Architecture, Engineering, and Construction (AEC industry) Development
- 13. Industrial Manufacturing & Machinery, Automation & Robotics, Consumer Electronics, and Energy
- 14. Semiconductor Development
- 15. AI in Product Development
- 16. Risk Management
- 17. Product Development Terms and Definitions
What is a Traceability Matrix? A Guide to Requirements Traceability
Every requirement in a complex product carries a history: where it came from, how it was built, and what proves it works. A requirements traceability matrix captures that story in one place, connecting each requirement to its source, its design, its tests, and its verification evidence. In regulated industries, auditors expect to follow that thread from need to proof, and teams that can’t show it usually don’t realize the gap until an audit surfaces it.
Below is a working template you can start from today, plus the context to build and maintain an RTM that actually holds up under change.
What is a Requirements Traceability Matrix (RTM)?
A requirements traceability matrix (RTM) is a document that maps every requirement in two directions. It traces backward to the stakeholder need or regulation that created it, and forward to the design elements, test cases, and verification activities tied to it. ISO/IEC/IEEE 29148, the international standard for requirements engineering, formalizes this definition.
How a Traceability Matrix Fits Into Requirements Management
Requirements management is how teams define, organize, and control what a product needs to do. The traceability matrix is the artifact that proves it actually happened. It connects each managed requirement to the design decisions, test cases, and verification records that address it, so nothing gets approved on paper without evidence behind it. Traceability works best as a daily habit, not a pre-audit sprint. Rebuilding links from memory under deadline pressure is how most gaps get introduced.
Why Traceability Matrices Matter
Traceability is a compliance expectation across medical devices, aerospace, automotive, and semiconductor development, and regulators can and do cite gaps. In medical devices, for example, design controls require traceable design inputs, outputs, verification, validation, and controlled changes.
The RTM gives teams a clear record of which requirement led to which work, and what evidence proves it was done correctly. That record pays off in four areas:
- Test coverage gaps: Forward traceability catches requirements that have no linked test cases. Backward traceability catches test cases or design elements that don’t trace to any requirement, which usually signals scope creep.
- Audit readiness: Auditors ask where a requirement came from and how you know it was satisfied. The RTM answers both. In September 2025, the FDA cited inadequate traceability between product requirements and risk management controls in a warning letter to Philips.
- Change impact: When a requirement changes mid-program, everything connected to it needs a second look. The RTM shows which design specs, code modules, tests, and risk controls are affected so the team knows what to reassess.
- Risk control: For medical devices and other risk-driven programs, teams extend the matrix to include hazards and risk controls so the safety case stays current when requirements change.
When trace links break in any of these areas, the result is usually rework, delayed submissions, or audit findings that could have been caught months earlier.
Types of Traceability Matrices
RTMs are built around several directions of traceability. Each one answers a different question about requirements coverage, and most programs need more than one working together.
Forward Traceability
Forward traceability runs from requirements down to design specs, implementation, and test cases. The question it answers is simple. “Have we built and verified everything we said we would?” In regulated programs, teams often treat it as a formal coverage metric.
Backward Traceability
Backward traceability works the other way. It starts from a design element, a test, or a piece of code and asks, “Why does this exist, and what requirement does it trace back to?” If something in the product can’t be linked to a requirement, it may be unintended behavior that was never reviewed for safety or compliance.
Bidirectional Traceability
Bidirectional traceability combines both directions and keeps the links consistent. It’s the only approach that supports both coverage analysis (requirements missing tests) and scope control (tests or design elements with no requirement). In practice, it requires distinct relationship types in each direction, because “verifies” is not the same relationship as “verified by.”
Horizontal Traceability
Horizontal traceability tracks dependencies across subsystems or disciplines at the same level of the architecture. In a complex product where mechanical, electrical, and software teams each own separate requirement sets, horizontal links show where one subsystem’s requirement depends on another’s. Without them, a change in one subsystem can break an assumption in another, and nobody finds out until integration.
Key Components of a Traceability Matrix
An RTM row should tell you the origin of a requirement, the design and code that implement it, and the test results that prove it works. The exact columns vary by standard and team workflow.
Core Matrix Elements
Every RTM row starts with a unique requirement identifier (for example, SYS-001 or HLR-042) that fits a consistent hierarchy. Bidirectional links connect the requirement upward to its source and downward to design specifications, code modules, and test cases.
Testing and Verification Fields
Test case IDs map to specific requirements for coverage analysis. Each mapped test records its verification method (test, inspection, analysis, or demonstration), execution status, and any linked defects. In risk-driven programs, the matrix also links hazards to risk controls and their verification evidence.
Status Tracking and Documentation
Regulated projects typically need change history, timestamps, and approval records to show who reviewed what and when. Spreadsheets can store this information, but they can’t enforce it. Jama Connect® maintains links, history, and approvals on the requirements and tests themselves, and its Live Traceability™ feature automatically flags every downstream item for review whenever an upstream requirement changes.
How to Create a Requirements Traceability Matrix
Whether you’re working in a spreadsheet or a dedicated tool, the process follows the same five steps. The audit trail looks very different depending on which you choose, but the logic is the same.
1. Identify and Baseline Your Requirements
Start by separating regulatory requirements (things like FDA design controls or DO-178C objectives) from your own internal product requirements, and then translate both into specific, testable statements. And baseline early, because without a locked revision, nobody can prove which version of the requirement the team actually verified.
2. Assign Unique IDs and Define Traceability Links
A hierarchical naming scheme works well. SYS-XXX for system-level requirements, HLR-XXX for high-level, and LLR-XXX for low-level. Standard link types like “derives-from,” “implements,” and “verifies” reduce ambiguity and determine what the team can report on later.
3. Map Requirements to Design, Tests, and Verification
The mapping approach varies by industry. FDA and ISO 13485 medical device teams commonly trace user needs to design inputs, then to design outputs, then to verification tests. DO-178C airborne software programs trace system requirements to high-level and low-level requirements, then to source code, with tests mapped at each level.
4. Validate Coverage and Close Gaps
Gap analysis has to run in both directions. Requirements with no downstream links mean something hasn’t been tested. But artifacts with no source requirement are just as concerning, because they usually signal scope creep or behavior that was never formally reviewed. In Jama Connect, teams model the expected relationships upfront and track exceptions during development.
5. Maintain and Update Throughout the Lifecycle
The matrix needs continuous maintenance, with updates tied to real changes rather than calendar milestones. When requirements change, the trace links identify all affected test cases, design documents, and code modules for reassessment. In a tool, the cycle is explicit. A requirement owner changes the text, downstream owners assess the flagged links, and the team re-verifies only what changed.
RELATED ARTICLE: Requirements Traceability Benchmark
Ready to Find Out More?
Our team of experts is here to answer any questions and learn how we can help enable your continued success. Get started with a free 30-day trial, or book a demo!
Traceability Matrix Example
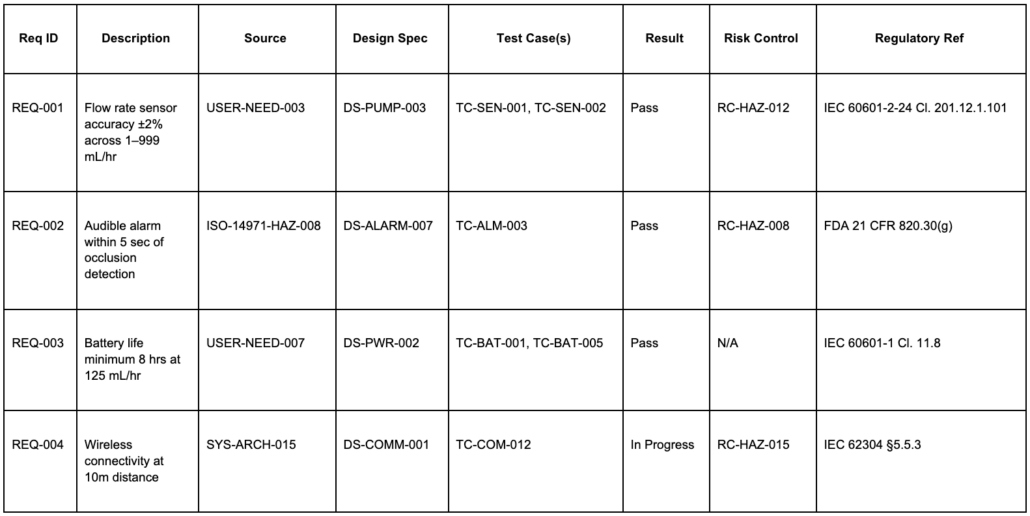
The table below shows four sample RTM rows for an FDA and IEC 62304-regulated infusion pump. Each row illustrates how a requirement connects to its source, design evidence, verification, and regulatory reference in one view. The column structure also works as a starting template for teams building their first requirements traceability matrix.
To see how this works in practice, take REQ-002. It traces backward to a hazard identified through ISO 14971 risk analysis (ISO-14971-HAZ-008) and forward to alarm design specification DS-ALARM-007, test case TC-ALM-003, and risk control RC-HAZ-008. If the occlusion detection algorithm changes, every linked artifact needs reassessment, including the test evidence and the risk control rationale. An auditor reviewing this row can verify the entire chain without chasing emails or cross-referencing disconnected documents.
Best Practices for Using a Traceability Matrix
Most traceability problems come down to two things: links that were never created, and links that weren’t updated when requirements changed. These best practices help prevent both.
Start Early and Keep It Simple
Traceability that begins at project kickoff costs far less than traceability reconstructed before an audit. A clear hierarchy and a small set of link types usually outperform a complex schema that nobody can maintain. The simplest check is whether every requirement has a source and at least one verification reference.
Use Bidirectional Traceability
Bidirectional traceability supports both coverage and scope control. Forward-only traceability can still leave orphaned tests or design elements that nobody can justify. Most workflows expect bidirectional traceability, and even when the standard doesn’t spell it out explicitly, auditors often sample artifacts in both directions.
Automate Where Possible
Manual traceability gets harder as systems grow and relationships multiply. You should aim to score requirement quality at authoring time, generate test cases directly from requirements, and maintain trace links automatically. Jama Connect Advisor™ does this as an add-on to Jama Connect, using AI to score requirement quality, flag ambiguities, and automatically generate test cases linked back to their source requirements.
Spreadsheets vs. Traceability Software
Spreadsheet-based RTMs and dedicated traceability software perform very differently during change and audit cycles. Spreadsheets can work early on, but they start breaking down once multiple teams and baselines are involved.
Common Pitfalls of Manual RTMs
Microsoft Word and Excel work for small teams with a handful of requirements. As requirements grow, spreadsheet maintenance becomes difficult to sustain. Here are the most common breakdowns:
- Version control breakdown: Multiple team members update the same file and copies circulate by email, so the team loses clarity on which version is authoritative.
- Absent audit trails: File timestamps show when a file changed, but not who changed which cell and why.
- Silent data corruption: An undocumented edit can invalidate coverage reports and delay a release.
- Part 11 friction: Spreadsheets don’t consistently enforce approvals, access controls, or electronic signature workflows expected in these environments.
These problems usually surface during a deadline or audit, when nobody has time to investigate.
What to Look for in RTM Software
When evaluating dedicated traceability software, the question is whether it can maintain links under change without turning traceability into a second job. These are the capabilities that teams in regulated industries should prioritize:
- Automated link handling: Baselines, version control, and change impact views keep links aligned as requirements evolve.
- Audit-ready history: The system records who changed what, when, and why, plus who approved it.
- Risk-aware trace chains: Regulated teams need trace links that connect hazards, controls, requirements, and verification evidence in a single chain. Jama Software’s risk management workflow is one example of this done natively.
- Bidirectional toolchain sync: When implementation work lives in separate systems, traceability breaks unless links stay current in both directions. Jama Software’s software development workflows maintain that connectivity across tools.
Once those capabilities are in place, the RTM becomes a byproduct of normal engineering work rather than a document rebuilt before each audit.
Why Most Traceability Problems Are Process Problems
Traceability failures are rarely caused by teams that don’t understand the concept. They happen when maintaining links takes too much effort, or the process makes it too easy to skip steps.
Jama Connect, a requirements management platform built for regulated product development, reduces that burden through Live Traceability. When a requirement changes, every linked design element, test case, and risk control is automatically flagged for review. Traceability Information Models (TIMs) define the expected relationships between requirements, designs, and tests, so missing links surface during development rather than during an audit.
If that’s the kind of visibility your team needs, start your free 30-day trial and explore how Jama Connect links requirements to verification evidence with a full audit trail.
Frequently Asked Questions About Traceability Matrices
What is the difference between a traceability matrix and a requirements traceability matrix?
The terms are often used interchangeably. Variations like Verification Cross-Reference Matrix (VCRM) or Requirements Verification Traceability Matrix (RVTM) emphasize different aspects but serve the same purpose. They all link requirements to their sources and to evidence of fulfillment.
Who is responsible for maintaining a traceability matrix?
Systems engineers typically own RTM creation and ongoing maintenance, but ownership spans multiple roles. Quality and regulatory affairs teams validate that the matrix meets audit requirements, and test engineers maintain verification links and results.
Can you use a traceability matrix in agile?
Yes, and most regulated teams use a hybrid approach. High-level requirements stay controlled while implementation and verification evolve across sprints. In practice, that means the RTM links from a stable high-level requirement to whichever sprint-level tests and design artifacts currently satisfy it, and those links update as each sprint delivers new verification evidence.
Can you manage a traceability matrix in Jira?
Jira tracks tasks and issues well, but it wasn’t built for requirements traceability in regulated environments. Teams that need formal trace links, baselines, and audit-ready evidence typically use a dedicated requirements tool alongside Jira. Jama Connect integrates with Jira through Jama Connect Interchange™, so engineering teams can work in Jira while requirements and traceability stay managed in Jama Connect.
How does a traceability matrix support compliance audits?
The RTM provides the chain of evidence auditors follow from stakeholder need through design, implementation, and verification. For FDA submissions, it supports the Design History File and design control evidence.
RELATED ARTICLE: BrightInsight Drives Efficiency Using Jama Connect®
In This Video, Learn About Live Traceability™ in Jama Connect®
A requirements traceability matrix (RTM) is a document or tool that maps project requirements to their corresponding test cases, ensuring that every requirement is addressed and validated.
Book a Demo
See Jama Connect in Action!
Our Jama Connect experts are ready to guide you through a personalized demo, answer your questions, and show you how Jama Connect can help you identify risks, improve cross-team collaboration, and drive faster time to market.