How to Write a System Requirements Specification (SRS) Document
The Essential Guide to Requirements Management and Traceability
Chapters
- 1. Requirements Management
- Overview
- 1 What is Requirements Management? A Complete Guide
- 2 Why do you need Requirements Management?
- 3 Four Stages of Requirements Management Processes
- 4 Adopting an Agile Approach to Requirements Management
- 5 Status Request Changes
- 6 Conquering the 5 Biggest Challenges of Requirements Management
- 7 Three Reasons You Need a Requirements Management Solution
- 8 Guide to Poor Requirements: Identify Causes, Repercussions, and How to Fix Them
- 2. Writing Requirements
- Overview
- 1 Functional requirements examples and templates
- 2 Identifying and Measuring Requirements Quality
- 3 How to Write a System Requirements Specification (SRS) Document
- 4 The Fundamentals of Business Requirements: Examples of Business Requirements and the Importance of Excellence
- 5 Adopting the EARS Notation to Improve Requirements Engineering
- 6 Jama Connect Advisor™
- 7 Frequently Asked Questions about the EARS Notation and Jama Connect Advisor™
- 8 How to Write an Effective Product Requirements Document (PRD)
- 9 Functional vs. Non-Functional Requirements
- 10 What Are Nonfunctional Requirements and How Do They Impact Product Development?
- 11 Characteristics of Effective Software Requirements and Software Requirements Specifications (SRS)
- 12 8 Do’s and Don’ts for Writing Requirements
- 3. Requirements Gathering and Management Processes
- Overview
- 1 Requirements Engineering
- 2 Requirements Analysis
- 3 A Guide to Requirements Elicitation for Product Teams
- 4 Requirements Gathering Techniques for Agile Product Teams
- 5 What is Requirements Gathering in Software Engineering?
- 6 Defining and Implementing a Requirements Baseline
- 7 Managing Project Scope — Why It Matters and Best Practices
- 8 How Long Do Requirements Take?
- 9 How to Reuse Requirements Across Multiple Products
- 4. Requirements Traceability
- Overview
- 1 How is Traceability Achieved? A Practical Guide for Engineers
- 2 What is Requirements Traceability? Importance Explained
- 3 Tracing Your Way to Success: The Crucial Role of Traceability in Modern Product and Systems Development
- 4 Bidirectional Traceability: What It Is and How to Implement It
- 5 What is Engineering Change Management (ECM)? A Complete Guide
- 6 Change Impact Analysis (CIA): A Short Guide for Effective Implementation
- 7 What is Meant by Version Control?
- 8 What is Requirements Traceability and Why Does It Matter for Product Teams?
- 9 Key Traceability Challenges and Tips for Ensuring Accountability and Efficiency
- 10 The Role of a Data Thread in Product and Software Development
- 11 Unraveling the Digital Thread: Enhancing Connectivity and Efficiency
- 12 What is a Traceability Matrix? A Guide to Requirements Traceability
- 13 How to Create and Use a Requirements Traceability Matrix (RTM)
- 14 Traceability Matrix 101: Why It’s Not the Ultimate Solution for Managing Requirements
- 15 Live Traceability vs. After-the-Fact Traceability
- 16 Overcoming Barriers to Live Requirements Traceability™
- 17 Requirements Traceability, What Are You Missing?
- 18 Four Best Practices for Requirements Traceability
- 19 Requirements Traceability: Links in the Chain
- 20 What Are the Benefits of End-to-End Traceability During Product Development?
- 21 FAQs About Requirements Traceability
- 5. Requirements Management Tools and Software
- Overview
- 1 Selecting the Right Requirements Management Tools and Software
- 2 Why Investing in Requirements Management Software Makes Business Sense During an Economic Downturn
- 3 Why Word and Excel Alone is Not Enough for Product, Software, and Systems Development
- 4 Can You Track Requirements in Excel?
- 5 Application lifecycle management (ALM)
- 6 Is There Life After DOORS®?
- 7 Can You Track Requirements in Jira?
- 8 Checklist: Selecting a Requirements Management Tool
- 6. Requirements Validation and Verification
- 7. Meeting Regulatory Compliance and Industry Standards
- Overview
- 1 Understanding ISO Standards
- 2 Understanding ISO/IEC 27001: A Guide to Information Security Management
- 3 What is DevSecOps? A Guide to Building Secure Software
- 4 Compliance Management
- 5 What is FMEA? Failure Mode and Effects Analysis Guide
- 6 TÜV SÜD: Ensuring Safety, Quality, and Sustainability Worldwide
- 8. Systems Engineering
- Overview
- 1 What is Systems Engineering?
- 2 How Do Engineers Collaborate? A Guide to Streamlined Teamwork and Innovation
- 3 The Systems Engineering Body of Knowledge (SEBoK)
- 4 What is MBSE? Model-Based Systems Engineering Explained
- 5 Digital Engineering Between Government and Contractors
- 6 Digital Engineering Tools: The Key to Driving Innovation and Efficiency in Complex Systems
- 9. Automotive Development
- 10. Medical Device & Life Sciences Development
- Overview
- 1 The Importance of Benefit-Risk Analysis in Medical Device Development
- 2 Software as a Medical Device: Revolutionizing Healthcare
- 3 What’s a Design History File, and How Are DHFs Used by Product Teams?
- 4 Navigating the Risks of Software of Unknown Pedigree (SOUP) in the Medical Device & Life Sciences Industry
- 5 What is ISO 13485? Your Comprehensive Guide to Compliant Medical Device Manufacturing
- 6 What You Need to Know: ANSI/AAMI SW96:2023 — Medical Device Security
- 7 ISO 13485 vs ISO 9001: Understanding the Differences and Synergies
- 8 Failure Modes, Effects, and Diagnostic Analysis (FMEDA) for Medical Devices: What You Need to Know
- 9 Embracing the Future of Healthcare: Exploring the Internet of Medical Things (IoMT)
- 11. Aerospace & Defense Development
- 12. Architecture, Engineering, and Construction (AEC industry) Development
- 13. Industrial Manufacturing & Machinery, Automation & Robotics, Consumer Electronics, and Energy
- 14. Semiconductor Development
- 15. AI in Product Development
- 16. Risk Management
- 17. Product Development Terms and Definitions
Chapter 2: How to Write a System Requirements Specification (SRS) Document
Chapters
- 1. Requirements Management
- Overview
- 1 What is Requirements Management? A Complete Guide
- 2 Why do you need Requirements Management?
- 3 Four Stages of Requirements Management Processes
- 4 Adopting an Agile Approach to Requirements Management
- 5 Status Request Changes
- 6 Conquering the 5 Biggest Challenges of Requirements Management
- 7 Three Reasons You Need a Requirements Management Solution
- 8 Guide to Poor Requirements: Identify Causes, Repercussions, and How to Fix Them
- 2. Writing Requirements
- Overview
- 1 Functional requirements examples and templates
- 2 Identifying and Measuring Requirements Quality
- 3 How to Write a System Requirements Specification (SRS) Document
- 4 The Fundamentals of Business Requirements: Examples of Business Requirements and the Importance of Excellence
- 5 Adopting the EARS Notation to Improve Requirements Engineering
- 6 Jama Connect Advisor™
- 7 Frequently Asked Questions about the EARS Notation and Jama Connect Advisor™
- 8 How to Write an Effective Product Requirements Document (PRD)
- 9 Functional vs. Non-Functional Requirements
- 10 What Are Nonfunctional Requirements and How Do They Impact Product Development?
- 11 Characteristics of Effective Software Requirements and Software Requirements Specifications (SRS)
- 12 8 Do’s and Don’ts for Writing Requirements
- 3. Requirements Gathering and Management Processes
- Overview
- 1 Requirements Engineering
- 2 Requirements Analysis
- 3 A Guide to Requirements Elicitation for Product Teams
- 4 Requirements Gathering Techniques for Agile Product Teams
- 5 What is Requirements Gathering in Software Engineering?
- 6 Defining and Implementing a Requirements Baseline
- 7 Managing Project Scope — Why It Matters and Best Practices
- 8 How Long Do Requirements Take?
- 9 How to Reuse Requirements Across Multiple Products
- 4. Requirements Traceability
- Overview
- 1 How is Traceability Achieved? A Practical Guide for Engineers
- 2 What is Requirements Traceability? Importance Explained
- 3 Tracing Your Way to Success: The Crucial Role of Traceability in Modern Product and Systems Development
- 4 Bidirectional Traceability: What It Is and How to Implement It
- 5 What is Engineering Change Management (ECM)? A Complete Guide
- 6 Change Impact Analysis (CIA): A Short Guide for Effective Implementation
- 7 What is Meant by Version Control?
- 8 What is Requirements Traceability and Why Does It Matter for Product Teams?
- 9 Key Traceability Challenges and Tips for Ensuring Accountability and Efficiency
- 10 The Role of a Data Thread in Product and Software Development
- 11 Unraveling the Digital Thread: Enhancing Connectivity and Efficiency
- 12 What is a Traceability Matrix? A Guide to Requirements Traceability
- 13 How to Create and Use a Requirements Traceability Matrix (RTM)
- 14 Traceability Matrix 101: Why It’s Not the Ultimate Solution for Managing Requirements
- 15 Live Traceability vs. After-the-Fact Traceability
- 16 Overcoming Barriers to Live Requirements Traceability™
- 17 Requirements Traceability, What Are You Missing?
- 18 Four Best Practices for Requirements Traceability
- 19 Requirements Traceability: Links in the Chain
- 20 What Are the Benefits of End-to-End Traceability During Product Development?
- 21 FAQs About Requirements Traceability
- 5. Requirements Management Tools and Software
- Overview
- 1 Selecting the Right Requirements Management Tools and Software
- 2 Why Investing in Requirements Management Software Makes Business Sense During an Economic Downturn
- 3 Why Word and Excel Alone is Not Enough for Product, Software, and Systems Development
- 4 Can You Track Requirements in Excel?
- 5 Application lifecycle management (ALM)
- 6 Is There Life After DOORS®?
- 7 Can You Track Requirements in Jira?
- 8 Checklist: Selecting a Requirements Management Tool
- 6. Requirements Validation and Verification
- 7. Meeting Regulatory Compliance and Industry Standards
- Overview
- 1 Understanding ISO Standards
- 2 Understanding ISO/IEC 27001: A Guide to Information Security Management
- 3 What is DevSecOps? A Guide to Building Secure Software
- 4 Compliance Management
- 5 What is FMEA? Failure Mode and Effects Analysis Guide
- 6 TÜV SÜD: Ensuring Safety, Quality, and Sustainability Worldwide
- 8. Systems Engineering
- Overview
- 1 What is Systems Engineering?
- 2 How Do Engineers Collaborate? A Guide to Streamlined Teamwork and Innovation
- 3 The Systems Engineering Body of Knowledge (SEBoK)
- 4 What is MBSE? Model-Based Systems Engineering Explained
- 5 Digital Engineering Between Government and Contractors
- 6 Digital Engineering Tools: The Key to Driving Innovation and Efficiency in Complex Systems
- 9. Automotive Development
- 10. Medical Device & Life Sciences Development
- Overview
- 1 The Importance of Benefit-Risk Analysis in Medical Device Development
- 2 Software as a Medical Device: Revolutionizing Healthcare
- 3 What’s a Design History File, and How Are DHFs Used by Product Teams?
- 4 Navigating the Risks of Software of Unknown Pedigree (SOUP) in the Medical Device & Life Sciences Industry
- 5 What is ISO 13485? Your Comprehensive Guide to Compliant Medical Device Manufacturing
- 6 What You Need to Know: ANSI/AAMI SW96:2023 — Medical Device Security
- 7 ISO 13485 vs ISO 9001: Understanding the Differences and Synergies
- 8 Failure Modes, Effects, and Diagnostic Analysis (FMEDA) for Medical Devices: What You Need to Know
- 9 Embracing the Future of Healthcare: Exploring the Internet of Medical Things (IoMT)
- 11. Aerospace & Defense Development
- 12. Architecture, Engineering, and Construction (AEC industry) Development
- 13. Industrial Manufacturing & Machinery, Automation & Robotics, Consumer Electronics, and Energy
- 14. Semiconductor Development
- 15. AI in Product Development
- 16. Risk Management
- 17. Product Development Terms and Definitions
How to Write a System Requirements Specification (SRS) Document
A well-written system requirements specification gives your entire team, from engineering to compliance, a shared foundation that keeps development on track and makes audits easier to pass. Getting the structure and content right from the start saves time at every stage that follows.
This guide covers what an SRS includes, how to write one that holds up under engineering scrutiny and regulatory audit, and where teams commonly introduce errors that compound downstream.
What Is a System Requirements Specification (SRS)?
A system requirements specification (SRS) is a formal document that captures every functional and non-functional requirement a system must satisfy. IEEE 830-1998 originally defined it as “a specification for a particular software product, program, or set of programs that performs certain functions in a specific environment.” The current standard, ISO/IEC/IEEE 29148, updated in 2018, expands that scope to cover the full requirements engineering lifecycle from elicitation through validation.
IEEE 830 organizes the SRS into three major sections:
- Introduction: Purpose, scope, definitions, acronyms, and references.
- Overall Description: Product context, major functions, user characteristics, constraints, and assumptions.
- Specific Requirements: All functional and nonfunctional requirements, external interface descriptions, design constraints, and quality attributes.
Design implementation details and project management content like cost or delivery schedules do not belong in the SRS. The document focuses on what the system must do, not how it will be built. That focus is what makes it useful across teams. Developers reference it for design, test engineers derive verification plans from it, and compliance officers use it to demonstrate regulatory conformance.
How an SRS Differs From Other Requirements Documents
ISO/IEC/IEEE 29148 defines three levels of requirements documents, each scoped to a different level of the system:
- Stakeholder Requirements Specification (StRS): Captures what stakeholders need the system to accomplish, written in their language.
- System Requirements Specification (SyRS): Translates those stakeholder needs into system-level requirements that can be allocated to hardware, software, and human elements.
- Software Requirements Specification (SRS): Specifies the software-level requirements in enough detail for developers to design and testers to verify.
Another common distinction is between an SRS and a functional requirements specification (FRS). An SRS covers both functional and non-functional requirements, while an FRS typically covers only the functional subset. For more on how these document types fit together, see the writing requirements chapter of the Jama Software requirements guide.
Why an SRS Matters for Product Development
Requirements problems found late in the development cycle are by far the most expensive to fix. An FDA study found that 44% of voluntary medical device recalls were preventable with adequate design controls, including proper requirements documentation. As software has become a larger share of medical device functionality, the problem has only grown since. Software design issues now account for 82% of all software-related recall causes.
The SRS is also where the verification chain starts. The FDA’s position is that software cannot be validated without predetermined, documented requirements. Without that documentation, teams have no objective basis for test planning and auditors have no traceable path to follow.
How to Write an SRS Step-by-Step
The following steps align with the IEEE 830 document structure and ISO/IEC/IEEE 29148’s requirements engineering lifecycle. They progress from scope definition through formal baseline approval.
1. Define Purpose, Scope, and Audience
The SRS should identify the software product by name, explain what the system will and will not do, and describe relevant benefits and objectives. It should also name the intended readers, including developers, testers, and project managers. The scope needs to be consistent with any higher-level system specification or StRS it traces to.
2. Document the Overall System Description
This section describes the general factors that affect the product and its requirements, including product context, major functions, user characteristics, and constraints covering regulatory, hardware, and safety considerations. Assumptions and dependencies should be documented explicitly so reviewers can evaluate whether they are still valid.
3. Write Functional Requirements
Functional requirements define the actions the system must perform, covering inputs, processing, and outputs. The EARS notation (Easy Approach to Requirements Syntax) provides six structured sentence patterns that replace vague natural language with testable statements. For example, “the system should handle orders properly” becomes “When an Order is shipped and Order Terms are not ‘Prepaid’, the system shall create an Invoice.”
4. Write Non-Functional Requirements
Performance, reliability, security, maintainability, and portability are common non-functional requirement categories. Every non-functional requirement must be quantifiable. “The system should be fast” fails the verifiability test, while “the website pages shall load within 3 seconds with the total number of simultaneous users below 5,000” can be measured and demonstrated to an auditor.
5. Define External Interfaces
The SRS must describe all inputs and outputs, including content, format, timing, valid ranges, and error handling. It should capture enough interface detail for independent design work and reference Interface Control Documents (ICDs) where applicable.
6. Set Acceptance Criteria and Traceability
Each requirement should be paired with a planned verification approach, whether that is examination, demonstration, analysis, or testing. Bidirectional traceability is mandatory in every regulated industry. Backward traceability links each SRS requirement to its origin in stakeholder needs, and forward traceability links each requirement to the design elements and test cases that address it. For a practical breakdown of why this matters in audits and change impact analysis, see end-to-end traceability.
7. Get Stakeholder Review and Approval
Once drafted, the SRS should go through formal review cycles. Many organizations use a System Requirements Review (SRR) as a gate for baseline approval. After the SRR, requirements go under configuration control, and any subsequent changes require formal impact assessment and approval by a Configuration Control Board (CCB).
Characteristics of a Well-Written SRS
IEEE 830 defines eight quality attributes for evaluating an SRS. The five most relevant for regulated product development are:
- Verifiable: A finite, cost-effective process exists with which a person or machine can check that the software product meets the requirement.
- Unambiguous: Every requirement has one and only one interpretation.
- Complete: The SRS includes responses to all realizable classes of input, including invalid inputs.
- Consistent: No requirements conflict with each other or with higher-level specifications.
- Traceable: The origin of each requirement is clear, and each requirement can be referenced in future development or verification documentation.
Auditors and reviewers evaluate SRS documents against these attributes during formal reviews, so they are worth keeping visible throughout the authoring process.
RELATED ARTICLE: The Easy Approach to Requirements Syntax (EARS)
Ready to Find Out More?
Our team of experts is here to answer any questions and learn how we can help enable your continued success. Get started with a free 30-day trial, or book a demo!
System Requirements Specification Example
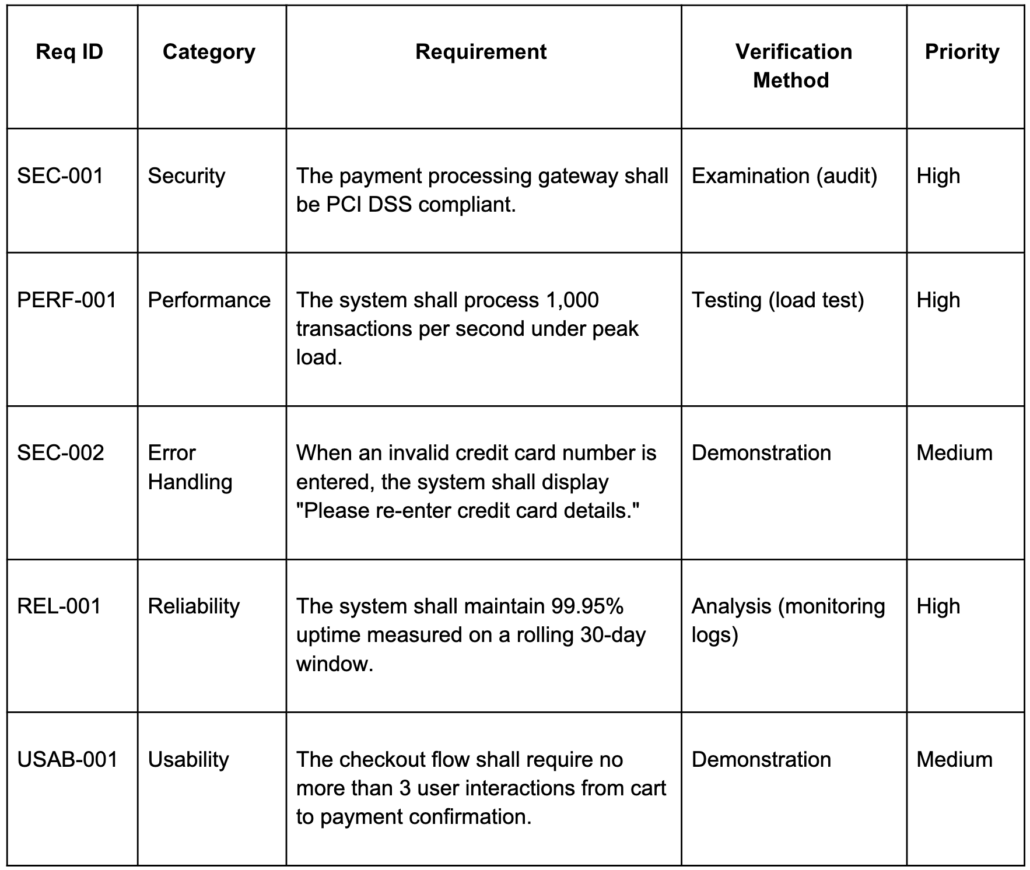
A vague requirement and a testable one can describe the exact same system — the difference is specificity and structure. Below is the same system described in two ways.
Vague requirements:
Rewritten using EARS patterns and IEEE 830 structure:
The vague versions fail IEEE 830’s verifiability test (“secure” and “real time” are subjective), are ambiguous (both terms can be interpreted multiple ways), and are incomplete (no specific error behavior defined). The rewritten versions add a unique ID, a category, a measurable threshold or concrete behavior, and a verification method so testers know exactly how to confirm each one.
SRS Document Template
The template below follows the IEEE 830 section structure and adds verification guidance from ISO/IEC/IEEE 29148. It can be adapted to meet industry-specific requirements like IEC 62304 for medical devices, DO-178C for aerospace, or ISO 26262 for automotive.
1. Introduction
- 1.1 Purpose
- 1.2 Scope
- 1.3 Definitions, Acronyms, and Abbreviations
- 1.4 References
- 1.5 Overview
2. Overall Description
- 2.1 Product Perspective
- 2.2 Product Functions
- 2.3 User Characteristics
- 2.4 Constraints
- 2.5 Assumptions and Dependencies
3. Specific Requirements
- 3.1 External Interface Requirements
- 3.2 Functional Requirements
- 3.3 Non-functional Requirements
- 3.4 Design Constraints
- 3.5 Quality Attributes
4. Verification Requirements (added by ISO/IEC/IEEE 29148)
5. Appendices (traceability matrices, glossary, interface control documents)
Sections 1 and 2 orient the reader and provide context. Section 3 is the core, where each requirement gets a unique ID, a “shall” statement, and a measurable acceptance criterion. Section 4 pairs every requirement with a verification method. Teams in regulated industries should also map each section to the relevant standard, such as IEC 62304 for medical device software or DO-178C for airborne systems.
Common Mistakes When Writing an SRS
Audit findings and post-release failure analyses across regulated industries point to the same recurring problems, and most of them start at the requirements stage:
- Vague or ambiguous language: Requirements that use subjective terms like “fast,” “user-friendly,” or “satisfactorily” cannot be tested and will not satisfy regulatory scrutiny. The FDA mandates each requirement to be evaluated for “accuracy, completeness, consistency, testability, correctness, and clarity.”
- Missing a glossary or data dictionary: IEEE 830 requires “definitions of all terms required to properly interpret the SRS.” When multi-meaning terms appear without glossary entries, different engineers reading the same requirement will reach different design conclusions.
- Mixing design decisions with requirements: A requirement that prescribes an implementation (such as “provide a database”) states a technology choice, not a need. When design decisions appear in the SRS, auditors question whether the team can trace requirements to genuine stakeholder needs.
- Skipping stakeholder sign-off: Proceeding to development without formal approval from quality, regulatory affairs, safety engineering, and clinical or business functions leaves the team without a defensible requirements baseline.
- Treating the SRS as a static document: The SRS must evolve as requirements change, risks surface, and regulations are updated. Formal version control and configuration management linking SRS versions to product builds are required in every regulated context.
These mistakes tend to compound: a vague requirement without a glossary entry gets interpreted differently by two engineers, and without stakeholder review, nobody catches the conflict until integration testing.
A Clear SRS Protects Your Program
Requirements problems that go undetected at the authoring stage compound through design, testing, and certification. Only 15% of teams use a dedicated requirements management solution, and poor requirements definition is a major cause of rework. As product complexity grows, manually maintaining requirements traceability across thousands of requirements and hundreds of test cases becomes unsustainable.
Jama Connect®, built for complex regulated product development, provides Live Traceability™ and Jama Connect Advisor™ to help teams write better requirements and maintain audit-ready traceability as their SRS evolves. Start your free 30-day trial today to see how it works for SRS development in your regulatory environment.
Frequently Asked Questions About System Requirements Specification Documents
What is the difference between an SRS and a product requirements document (PRD)?
An SRS is a formal document type defined by IEEE 830 and ISO/IEC/IEEE 29148, with mandatory sections and verification requirements. A PRD is a product management artifact focused on features and market requirements. It is not tied to any systems engineering standard and does not carry the contractual or legal weight an SRS carries in regulated environments.
How long should an SRS document be?
Length depends on system complexity, not on a target page count. IEEE 830 does not prescribe length. It prescribes completeness, meaning every significant requirement, including responses to invalid inputs, must be documented.
Can you use an SRS in agile development?
Yes. Teams using agile approaches treat the SRS as living documentation and maintain compliance metrics continuously at each iteration. For more on how this works in regulated environments, see the Traceable Agile approach.
What standard governs SRS document format?
IEEE 830-1998 was the original SRS standard, but it has been officially superseded by ISO/IEC/IEEE 29148:2018. New projects should use ISO/IEC/IEEE 29148 as their foundation, with industry-specific overlays (IEC 62304, DO-178C, ISO 26262) applied based on the applicable regulatory environment.
RELATED ARTICLE: The Easy Approach to Requirements Syntax (EARS)
In This Webinar, We Offer Best Practices for Writing Requirements
System Requirement Specification (SRS): The SRS is focused on what the software needs to do and how it must perform. It lays the important groundwork so that every person involved with the project understands the most crucial details.
Book a Demo
See Jama Connect in Action!
Our Jama Connect experts are ready to guide you through a personalized demo, answer your questions, and show you how Jama Connect can help you identify risks, improve cross-team collaboration, and drive faster time to market.