ISO 26262 vs. ASPICE for Automotive Compliance
Of course, automotive companies already use ISO 26262, and introducing yet another automotive compliance piece into a very full process may feel overwhelming. It’s understandable why companies would be asking if they need to adhere to both ASPICE and ISO 26262 when they are already focused on ISO 26262 compliance.
The answer, in short, is that while there is no regulatory requirement to use ASPICE, using the model can greatly benefit companies that want to stay competitive in the automotive industry. According to the Project Management Institute, 47% of project failures can be traced back to poor requirements; any guidance or set of standards that can help mitigate that risk is worth the implementation effort.
While ASPICE and ISO 26262 are complementary and do overlap in places, they ultimately serve different purposes. ISO 26262 covers functional safety standards for vehicles. It incorporates safety analysis methods that account for random and systematic errors in electrical and electronic systems and is broadly adopted worldwide. ASPICE is the current standard for software best practices in the automotive industry. It covers how to conduct software and systems design whether or not safety is a concern.
The best approach for automotive development teams is to consider both ASPICE and ISO 26262 guidelines. Below we will give a brief overview of both standards and discuss the similarities and differences.
ISO 26262 Explained
ISO 26262, titled “Road vehicles – Functional safety,” is an international standard for the functional safety of electrical and electronic (E/E) systems within road vehicles. Originating from the more generic IEC 61508 standard for electrical/electronic/programmable electronic safety-related systems, ISO 26262 addresses the specific needs and challenges of automotive E/E systems safety lifecycle management. This standard aims to ensure that E/E systems in vehicles are designed and developed to meet stringent safety requirements, reducing the risk of failures that could lead to accidents and harm.
The ISO 26262 standard is structured into several parts, covering aspects such as vocabulary, management of functional safety, concept phase, product development at the system, hardware, and software levels, production, operation, service, and decommissioning. It also includes guidance on automotive safety integrity levels (ASILs), which are used to classify and manage the safety requirements necessary to mitigate risks to an acceptable level.
Key aspects of ISO 26262 include:
- Risk Analysis and Management: It emphasizes the identification, evaluation, and mitigation of risks associated with E/E system failures throughout the vehicle’s lifecycle.
- Systematic and Random Hardware Failures: The standard addresses both systematic failures (due to errors in specification, design, manufacture, etc.) and random hardware failures, proposing methods to manage and mitigate their effects.
- Functional Safety Assessment: It requires a structured functional safety assessment to be conducted at various stages of the product development process, ensuring that all safety goals have been met.
- Automotive Safety Integrity Levels (ASILs): ISO 26262 introduces ASILs, which are assigned based on the severity, exposure, and controllability of potential hazards. ASILs range from A (lowest) to D (highest), dictating the rigor of safety measures needed.
- Safety Lifecycle: The standard outlines a safety lifecycle for the development of automotive E/E systems, including specific processes and tasks that must be followed to achieve functional safety.
- Documentation and Evidence: Comprehensive documentation and evidence of compliance with the standard’s requirements are critical for the certification process, supporting the safety case of the E/E system.
ISO 26262 is applicable to all types of passenger cars, motorcycles, trucks, buses, and trailers, with its principles also being adapted for use in other automotive applications. The standard is continually evolving to address the advancements in automotive technologies, such as autonomous vehicles and electric mobility, ensuring it remains relevant and effective in managing functional safety in the dynamic automotive industry.
ASPICE Explained
Automotive SPICE (Software Process Improvement and Capability dEtermination) is a framework used within the automotive industry to assess and improve the maturity of software development processes. It is based on the ISO/IEC 15504 standard, often referred to as SPICE, and tailored specifically for automotive software development and related system integration processes. The framework is designed to help organizations develop high-quality automotive software more efficiently, ensuring that it meets both customer expectations and regulatory requirements.
ASPICE provides a structured approach to evaluating the capability levels of an organization’s processes in a consistent manner. It defines a set of process assessment models and practices that organizations can use to measure their processes against industry best practices. The framework focuses on key process areas such as software engineering, project management, quality assurance, and supplier management.
Key features of ASPICE include:
- Process Reference Model (PRM): This model defines the processes considered essential for the development and management of automotive software. Each process is described in terms of its purpose, outcomes, and outputs.
- Process Assessment Model (PAM): The PAM provides criteria for assessing the maturity levels of the processes defined in the PRM. It outlines capability levels (ranging from 0 to 5) and process attributes that are used to evaluate the performance and capability of processes.
- Capability Levels: These levels describe the maturity and capability of processes within an organization. They range from Level 0 (Incomplete) to Level 5 (Optimizing), with higher levels indicating more mature and capable processes.
- Assessment and Improvement: ASPICE not only enables the assessment of current process capabilities but also provides a framework for continuous process improvement. Organizations can identify gaps in their processes and implement targeted improvements to enhance their software development capabilities.
ASPICE assessments are typically conducted by certified assessors who evaluate an organization’s processes against the framework’s criteria. The outcome of an assessment can help organizations identify areas for improvement, increase the efficiency of their software development processes, and enhance the quality of their automotive software products.
By implementing ASPICE, organizations in the automotive industry can achieve several benefits, including improved process transparency, higher software quality, reduced development risks, and better alignment with industry best practices. As automotive systems become increasingly software-driven, adhering to frameworks like ASPICE is becoming more critical for manufacturers and suppliers aiming to meet the high safety, reliability, and performance standards expected in the industry.
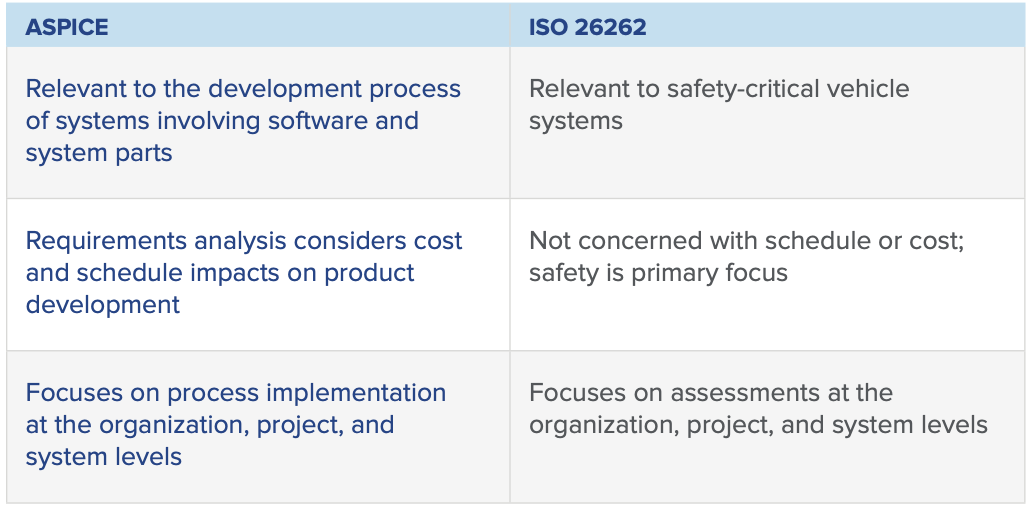
ISO 26262 vs. ASPICE: Similarities and Differences
There are several key distinctions between ASPICE and ISO 26262:
Stay tuned for our next post in the ASPICE 101 blog series where we discuss goals, requirements, and levels of ASPICE compliance.
Editors note: This post was written partially assisted by artificial intelligence. It was reviewed for accuracy by McKenzie Jonsson and Deco Wilkerson.
- Bosch and the Chiplet Revolution: Enabling Software-Defined Mobility - February 24, 2026
- Requirements Management Software Jama Connect Breaks Records for Scalability - February 9, 2026
- Next Generation Nuclear: Reactor Innovations Shaping 2025 - November 11, 2025