Live Requirements Traceability
Achieving Live Traceability™ of product requirements, as necessitated by industry standards, across siloed engineering teams and tools, is the #1 unsolved problem for most product development organizations. One of the main barriers is that each engineering discipline (systems, software, hardware, electrical, risk, verification and validation) has optimized its own process and tools. When looking at the end-to-end product development process siloed teams, tools, and data make it very challenging to trace development activity from initial requirement definition through development and testing.
As a result, requirements traceability becomes a time-consuming, error-prone, frustrating, and manual, after-the-fact process. The inability for the product development organization to continually trace ongoing development efforts and changes back to user and system requirements results in missed requirements, defects, rework, delays, audit letters, and cost overruns.
RECOMMENDED READING: Requirements Traceability: How to Go Live
No common platform exists
The typical approach to solve this generic process problem with software is to force every user onto a single platform and follow one common process. This works for standard business processes in HR, Sales, and Finance, but engineering disciplines across systems, software, hardware, electrical, risk, test, verification, and validation each follow different methodologies and use multiple tools including spreadsheets, desktop, and homegrown applications. Each engineering discipline has optimized their own development environment and strongly resist any attempts to change. Engineering leadership defers to each engineering discipline to define how to best do their work and is loath to dictate processes and tools that will negatively impact the performance and morale of each engineering team.
In addition to the organizational barriers to standardization, no single platform is even close to currently existing which replaces these dozens of tools. A single platform would need to cover all of the following software categories AND address all functionality in spreadsheets (Excel), scripts, desktop, and homegrown tools: Requirements Management, CAD, MBSE, DFMEA/FMEA, software task management, software code management, automated software testing, hardware bench test tools, ALM, PLM, and more. Current efforts by legacy vendors to create a common SaaS platform to span all these software categories and reach parity with best-of-breed tools is moving very slowly.
RECOMMENDED READING: Benefits of End-to-End Traceability
How to achieve Live Traceability™ without forcing change on engineering teams
So how does an organization achieve Live Traceability across a best-of-breed tool environment supporting disparate methodologies, terminologies, fields, and statuses? The answer is a 3-step approach:
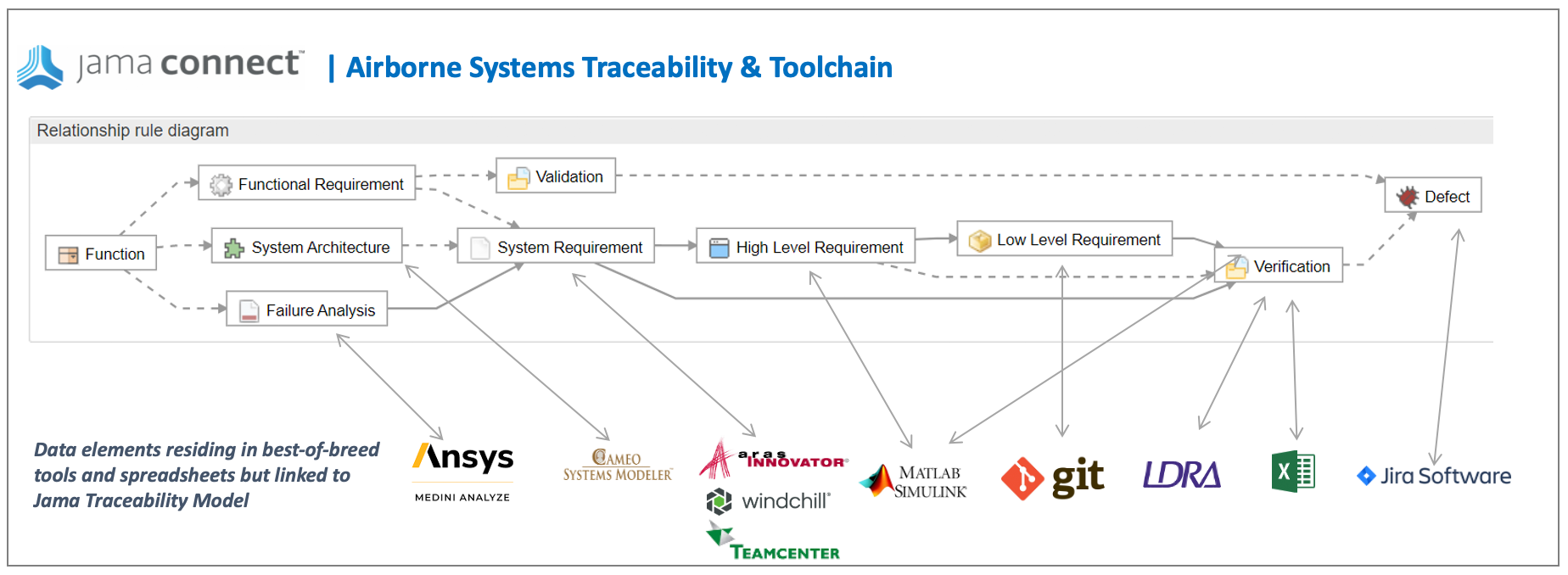
Step One | Live Traceability Model
Define a Live Traceability model across the end-to-end product development process with relationship rules for the traceable data elements across best-of-breed tools. An automotive functional safety example is shown below. Here you can see the operational instantiation of functional safety standards requirements in a relationship model within Jama Connect®. All necessary traceable information is included with continuous syncs to best-of-breed tools within engineering teams to deliver Live Traceability.
Step Two | Adaptive Data Field Mapping
To achieve Live Traceability, integrations with best-of-breed tools (such as those shown in the example) are required. The typical integration approach standardizes field names and statuses to ensure consistency across the connected tool, but this does not achieve the dual objective of Live Traceability with no changes required in how each engineering team works. Alternately, the proven approach is to apply adaptive data field mapping to ensure no change to engineering teams’ fields and statuses which simultaneously ensures a consistent, process wide, Live Traceability model. This is achieved through robust mapping and normalization logic functionality to easily address the various approaches taken by each engineering team.
RECOMMENDED READING: Requirements Management Guide: Requirements Traceability
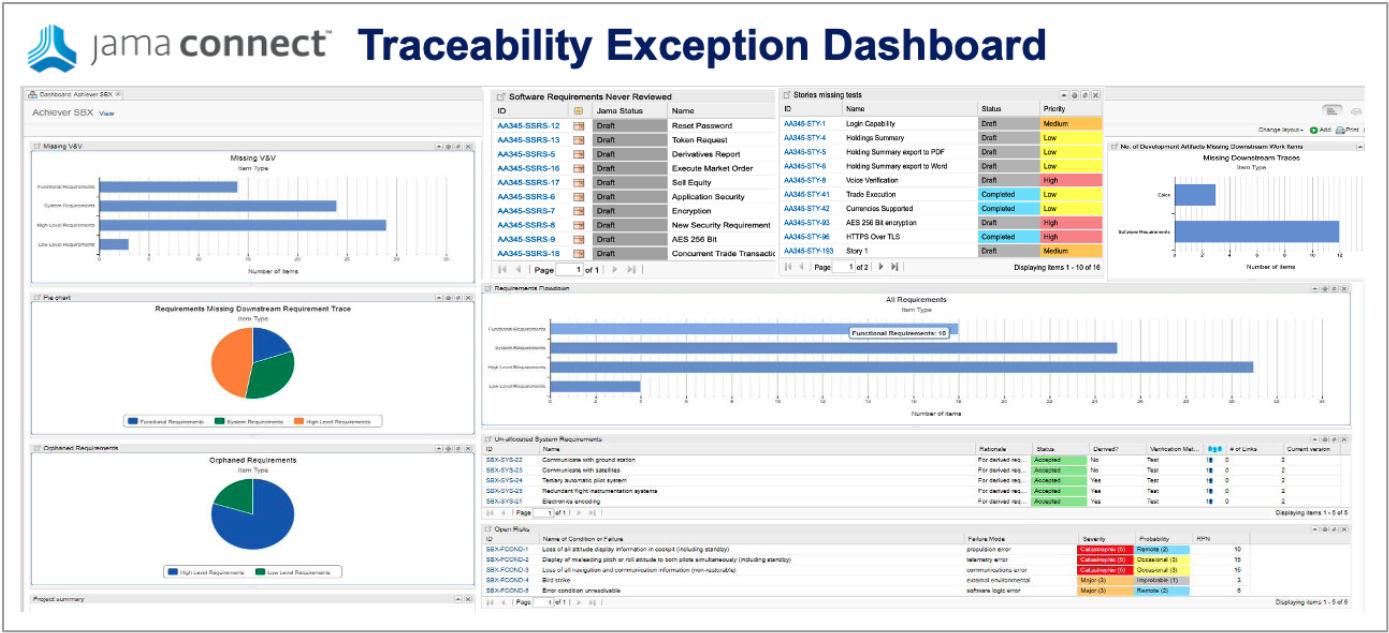
Step Three | Management by Exception
Once Live Traceability is achieved, engineering organizations can — for the first time — manage the end-to-end product development process in real time, identify exceptions to the process early, and take action to significantly reduce defects, rework, delays, and cost overruns.
LEARN MORE




![[Webinar Recap] Best Practices for Requirements Traceability [Webinar Recap] Best Practices for Requirements Traceability](https://www.jamasoftware.com/media/2021/10/WB-best-practices-traceability_social-Image.png)


